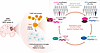Commentary
Abstract
Cytosolic DNA sensing through the cyclic GMP-AMP synthase (cGAS)–stimulator of interferon genes (STING) pathway has emerged as a promising strategy to elicit antitumor immunity. However, clinical translation of STING agonists has been hindered by limited efficacy and dose-limiting inflammatory toxicity, highlighting that simply providing activating ligands is insufficient to achieve durable immune responses. In this issue of the Journal of Clinical Investigation, Liao et al. showed that intracellular aspartate availability critically shapes STING signaling responsiveness. Aspartate deficiency disrupted pyrimidine synthesis, induced mtDNA stress, and engaged a feed-forward Z-DNA binding protein 1 and receptor interacting serine/threonine kinase 1/3 axis. Rather than directly triggering immunity, this metabolic state primed DNA sensing and fueled downstream signaling, thereby enabling robust and sustained antitumor immune responses. Together, these findings position nucleotide metabolism as a key determinant of innate immune responsiveness and suggest that metabolic conditioning may enhance the efficacy of STING-targeted therapies.
Authors
Haitao Jiang, Wenyan Wang, Yang-Xin Fu
Abstract
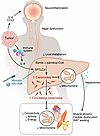
Liver fibrosis is a common pathological outcome of chronic liver disease and is driven by inflammatory responses. However, the early signals that initiate the inflammatory cascade remain poorly understood. Emerging evidence suggests that liver sinusoidal endothelial cells (LSECs) are not merely passive bystanders, but active regulators during liver fibrosis. In this issue of the JCI, Gan et al. demonstrated in multiple preclinical models that BRD4/PML-mediated super-enhancer activation in LSECs drives proinflammatory angiocrine signaling, thereby initiating liver fibrosis. Thus, targeting this endothelial axis may offer a promising therapeutic strategy for the treatment of liver fibrosis.
Authors
Yingfen Chen, Yong He
Abstract
The Western diet (WD) is a rich source of saturated fatty acids, especially palmitic acid (PA), which has been implicated in the pathogenesis of insulin resistance, oxidative stress, inflammation, diabetes, and multiorgan dysfunction in obesity and diabetes. In this issue of the JCI, a study by Balasubramaniam et al. describes mechanisms linking a WD, PA, ferroptosis (iron-dependent cell death), and loss of colonic motility. Chronic PA exposure drove ferroptosis in murine in vitro systems and human myenteric ganglia. Mice fed a WD for 12 weeks developed enteropathy and loss of colonic motility, which was reversed by adeno-associated virus–mediated (AAV-mediated) overexpression of the transcription factor NFE2L2, preventing ferroptosis and restoring redox balance to enteric neurons. The study provides critical data establishing PA-induced ferroptosis as a mediator and potential therapeutic target in enteric nervous system disorders associated with obesity.
Authors
Rexford S. Ahima
Abstract
Cancer cachexia, characterized by weight loss, muscle wasting, and anorexia, complicates cancer treatment and adversely affects patient outcomes. Both tumor-derived and host inflammatory factors are implicated in aspects of cachexia. The search for circulating mediators of cancer cachexia has focused largely on secreted proteins, but metabolites may also drive systemic wasting. In this issue, Morigny, Rohm, and colleagues identified the liver as a major source of circulating ceramides in cachectic mice and patients with cancer and demonstrated that inhibiting ceramide synthesis attenuated muscle wasting and preserved function in cachectic mice. These findings position the liver as an endocrine organ in cachexia and introduce a druggable metabolic pathway with translational potential.
Authors
Kerui Huang, Norbert Perrimon, Marcus D. Goncalves
Abstract
Glioma stem cells (GSCs) are a small subset of self-renewing, plastic, and multipotent neoplastic cells in glioblastoma (GBM) that sit at the apex of a cellular differentiation hierarchy. Elucidating pathways that enhance GSC properties and determine their cell-specific interactions within the immunosuppressive GBM microenvironment are critical for developing effective therapeutic approaches. The CLOCK-BMAL1 complex, which is well known for its activity as a circadian rhythm–regulating transcription factor, plays a critical role in maintaining GSC stemness, and the gene encoding CLOCK was found to be amplified in about 5% of GBM cases. Here, Zhou et al. have uncovered a “symbiotic exclusivity” relationship between CLOCK-BMAL1 and TFPI2, which is also amplified in a small proportion of GBM cases. This relationship forms a HIF-1α/NF-κB P65–mediated positive feedback loop that boosts the proliferative and tumor-enhancing capacities of GSC and immunosuppressive microglia. This self-amplifying regulatory circuit represents an opportunity for intervention to inhibit GBM growth.
Authors
Petros Basakis, Ling-kai Shih, Jiabo Li, Daniel J. Brat
Abstract
Clinical management of pancreatic cancer (PC) remains severely limited, primarily due to the complex tumor microenvironment. Emerging DNA damage–targeted strategies have demonstrated considerable therapeutic potential in PC. In this issue of the JCI, Teh et al. employed cancer-specific multitarget sgRNAs to induce DNA double-strand breaks (DSBs), resulting in lethal effects in PC cells. Integrative bioinformatic and cytogenetic analyses revealed that CRISPR/Cas9-mediated DSBs provoked persistent chromosomal instability, ultimately leading to chromosome catastrophe and cell death. Compared with equivalent radiation-induced DSBs, these sgRNAs exhibited superior cytotoxicity and were able to eliminate cells resistant to a specific sgRNA via subsequent targeting at distinct genomic sites, highlighting a promising and innovative precision therapeutic approach for clinical treatment of PC.
Authors
Li-Chan Chang, Christine E. Eyler, Chang-Lung Lee
Abstract
Immune checkpoint inhibitors (ICIs) have improved patient outcomes substantially in non–small cell lung cancer (NSCLC). Despite considerable effort, our understanding of the features that predict for immunotherapy response and resistance in patients remains incomplete. In this issue of the JCI, Isomoto and colleagues utilized a multiplex IHC platform to profile the spatial organization of the lung cancer tumor immune microenvironment, enabling the identification of spatial immune features that correlate with immunotherapy efficacy. This study enhances our knowledge of the spatial organization of features impacting ICI efficacy by identifying a three-variable spatial composite — including CD73 upregulation in EGFR-mutant NSCLC — that substantially outperforms PD-L1 expression in predicting immunotherapy efficacy. Moreover, it establishes spatial proteomic profiling as a platform for generating therapeutic hypotheses that are actionable and mechanistic in NSCLC.
Authors
Tao Zou, John D. Minna
Abstract
Despite extensive advances in understanding sepsis pathophysiology, treatment outcomes have not substantially improved. In this issue, Takahama and colleagues identified phospholipase A2 Group V (PLA2G5) as a contributor to sepsis lethality in mouse models of endotoxemia and sepsis. Whole-mouse spatial profiling generated bodywide maps of systemic inflammation and uncovered intestinal goblet cells as a source of pathogenic PLA2G5. Pairs of inflammatory cytokines (TNF and IFN-γ, or TNF and IL-18) induced PLA2G5 expression in goblet cells. Mechanistically, circulating PLA2G5 triggered intravascular hemolysis through its lipolytic activity on erythrocyte membranes and contributed to organ failure and death. PLA2G5’s deleterious effects were blocked by specific antibodies and were absent in Pla2g5-deficient mice. In humans with bacterial or fungal sepsis or severe COVID-19, plasma PLA2G5 levels were elevated and predicted disease severity. This discovery highlights the contribution of hemolysis to sepsis, suggesting that PLA2G5 inhibitors, hemoglobin, or heme antagonists could represent valuable therapeutic tools.
Authors
Jean-Marc Cavaillon
Abstract
Fatty acid oxidation (FAO) provides the healthy heart with 60%–90% of its ATP, with the remainder coming from metabolism of glucose. Metabolic flexibility is key to heart function, ensuring an uninterrupted source of fuel. In heart failure, a shift from FAO to glucose-dependent metabolism occurs as disease progresses, supporting the widely held notion that fat is the optimal substrate in the heart. In this issue of the JCI, Kim et al. challenge this assumption. In studies of acetyl-CoA carboxylase–deficient (ACC-deficient) mice, they found that unregulated use of fat as a substrate led to cardiac damage. ACC-deficient mice developed cardiolipin deficiency as a result of excessive FAO depleting stores of linoleic acid, which is used as a substrate for cardiolipin maturation. The resulting mitochondrial dysfunction was associated with dilated cardiomyopathy and heart failure in these mice. The findings highlight potential for development of therapeutic strategies that balance energy sources and replenish cardiolipin levels.
Authors
Steven M. Claypool, Carla M. Koehler
Abstract
Chemotherapy-induced alopecia (CIA) is a common and highly visible adverse effect of chemotherapy with substantial psychosocial and quality-of-life burdens. In this issue, Gherardini and colleagues described a targeted strategy to prevent CIA using ALRN-6924, a stapled peptide that transiently activates p53 and induces cell cycle arrest in proliferating TP53 wild-type tissues, such as the hair follicle. In ex vivo human scalp hair follicle culture, ALRN-6924 protected matrix keratinocytes and bulge stem cells from paclitaxel- and cyclophosphamide-induced injury, reducing apoptosis, DNA damage, and other pathologic features. These findings nominate precision chemoprotection as a promising supportive care approach for mitigating CIA.
Authors
Edward B. Li, Meredith Klay, Rui Yi
No posts were found with this tag.



Copyright © 2026 American Society for Clinical Investigation
ISSN: 0021-9738 (print), 1558-8238 (online)