Advertisement
Review Series
Open Access |  10.1172/JCI199850
10.1172/JCI199850
Immune signaling and function in neurodegeneration
Yvonne L. Latour and Dorian B. McGavern
Viral Immunology and Intravital Imaging Section, National Institute of Neurological Disorders and Stroke (NINDS), National Institutes of Health (NIH), Bethesda, Maryland, USA.
Address correspondence to: Dorian B. McGavern, National Institutes of Health, Building 10, Room 5N240C, Bethesda, Maryland 20892, USA. Phone: 301.443.7949; Email: mcgavernd@mail.nih.gov.
Find articles by Latour, Y. in: PubMed | Google Scholar
Viral Immunology and Intravital Imaging Section, National Institute of Neurological Disorders and Stroke (NINDS), National Institutes of Health (NIH), Bethesda, Maryland, USA.
Address correspondence to: Dorian B. McGavern, National Institutes of Health, Building 10, Room 5N240C, Bethesda, Maryland 20892, USA. Phone: 301.443.7949; Email: mcgavernd@mail.nih.gov.
Find articles by
McGavern, D.
in:
PubMed
|
Google Scholar
|

Published April 15, 2026 - More info
J Clin Invest. 2026;136(8):e199850. https://doi.org/10.1172/JCI199850.
© 2026 Latour et al. This work is licensed under the Creative Commons Attribution 4.0 International License. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
-
Abstract
Neurodegenerative diseases arise from interactions among pathogenic proteins, immune responses, and diverse environmental or age-related stressors that disrupt CNS homeostasis. CNS resident microglia detect self-derived danger signals through pattern recognition receptors, and their activation can promote clearance of aberrant proteins, including amyloid-β, tau, α-synuclein, and TAR DNA-binding protein 43. However, microglial activation may also drive maladaptive states that amplify neuroinflammation. Microglial transitions are further shaped by receptor-mediated signaling and antigen presentation pathways that integrate environmental cues with functional responses. Adaptive immune cells contribute additional layers of regulation, with CD8+ and CD4+ T cells exerting neuroprotective or neurotoxic effects depending on disease context, activation state, and antigen specificity. The identification of granzyme K–expressing CD8+ T cells in several neurodegenerative conditions highlights the growing recognition that distinct T cell subsets may have specialized roles in disease. Aging, repetitive head injury, and viral infection further alter microglial phenotypes, weaken barrier integrity, promote T cell recruitment, and prime the CNS for chronic inflammation. In this review, we synthesize current knowledge of innate and adaptive immune mechanisms in neurodegeneration, examine how external factors influence these responses, and consider how these insights may guide future therapeutic strategies.
-
Introduction
Neurodegenerative diseases remain a major clinical concern, affecting millions of individuals worldwide. Although a wide range of disorders fall under the neurodegenerative disease umbrella, many share hallmark features, including neuronal cell death, cognitive dysfunction, and aberrant protein accumulation, which have been reviewed elsewhere (1). Our understanding of the pathophysiology of Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), Lewy body dementia (LBD), amyotrophic lateral sclerosis (ALS), and frontotemporal dementia (FTD) has expanded beyond the classical association of neuronal loss and immune reactivity localized to regions of protein aggregation. This shift has been driven by the availability of new research tools and approaches (2–7) that have revealed the immune system not only as a key driver of neurodegenerative disease progression but also as a contributor to disease onset through maladaptive cellular activation and signaling (2–4, 8).
Under steady-state conditions, the CNS parenchyma relies on resident immune cells to maintain the blood–brain barrier (BBB), survey the local environment, and respond to cellular damage, pathogens, or dysfunctional cells. Neurons activate intrinsic signaling pathways in response to cellular dysfunction, while astrocytes and microglia are rapidly engaged to mitigate danger signals and, when necessary, recruit peripheral immune cells. During neurodegeneration, however, these responses can exacerbate aberrant protein deposition, and the balance of beneficial versus detrimental immune involvement remains incompletely understood. In this review, we explore key signaling pathways, cellular players, and immune responses that are shared across multiple neurodegenerative diseases, and we discuss how additional factors such as aging, infection, and repetitive injury may influence disease progression within the CNS.
-
Innate immune signaling
The innate immune response has evolved mechanisms to detect both externally derived stimuli, known as pathogen-associated molecular patterns (PAMPs), and endogenous molecules released from stressed or dying cells, referred to as damage-associated molecular patterns (DAMPs) (Figure 1A). These signals are recognized through pattern recognition receptors (PRRs) (9–11). As the primary resident immune cells of the CNS, microglia express high levels of PRRs; however, evidence also supports PRR expression on other CNS cell types, including astrocytes, oligodendrocytes, endothelial cells, and neurons (12–16).
 Figure 1
Figure 1Innate immune activation and microglial transition during neurodegeneration. (A) During neurodegeneration, distressed neurons release DAMPs such as mtDNA, ROS, HMGB1, aberrant proteins, and pTau–bound RNA. These signals activate resident microglia and astrocytes, which then recruit peripheral immune cells. As shown in more detail in B, activated microglia upregulate PRRs, including TLRs, RAGE, and cGAS, enabling detection of neuron-derived DAMPs. Engagement of PRRs induces proinflammatory gene expression programs and activates inflammasome pathways that promote release of chemokines and cytokines into the CNS parenchyma. As microglia transition from homeostatic to reactive states in a TREM2-dependent manner, damage-associated microglia increase MHC-II expression to support antigen presentation and coordination of adaptive immune responses that attempt to limit neurodegeneration.
TLRs. The most well-studied family of PRRs are the TLRs, first identified and characterized for their ability to sense a variety of microbial components (Figure 1B) (17–19). TLRs are transmembrane proteins that contain a leucine-rich repeat (LRR) extracellular domain and a cytosolic Toll IL-1 receptor (TIR) domain responsible for downstream signaling (18). When TLRs bind PAMPs, the intracellular domain recruits the adaptor proteins myeloid differentiation factor 88 (MyD88) or Tir domain–containing adaptor inducing interferon β (TRIF). This process results in the nuclear translocation of transcription factors such as NF-κB, which initiates proinflammatory gene expression programs (18, 20). Later studies showed that TLR activation can also occur in response to DAMPs during sterile inflammation in the CNS (21).
As a primary hallmark of neurodegenerative diseases, aberrant proteins such as amyloid β (Aβ), phosphorylated tau (pTau), α synuclein (α-syn), and TAR DNA binding protein 43 (TDP-43) have been identified as major DAMPs that activate PRRs in the CNS (22, 23), and these proteins are sometimes complexed with nucleic acids like RNA (Figure 1B) (24–29). Plasma membrane–bound TLR2 and TLR4 are upregulated in the brains of patients with AD and in transgenic AD mouse models (30). Binding of Aβ to TLR2 and TLR4 increases expression of immunomodulatory cytokines, including IL-1β, IL-6, IL-10, IL-17, and TNFα, in microglia and astrocytes (31–34). Genetic ablation or pharmacologic blockade of TLR2 and TLR4 exacerbate cognitive decline and increase Aβ burden in the brains of 5xFAD and APP/PS1 mice by reducing microglial activation (32, 34–36). However, an appropriate balance must be maintained, since persistent activation of the proinflammatory response downstream of TLR4 can be detrimental and amplify neurotoxicity (37, 38). In addition, recognition of Aβ by TLR4 expressed on neurons induces apoptosis and contributes to neurodegeneration (16). Similar mechanisms have been observed in other neurodegenerative disease models. Extracellular pathogenic α-syn, superoxide dismutase 1 (SOD1), and tau released by damaged neurons activate TLR2 and TLR4 and promote microglial reactivity in vitro and in vivo in models of PD, ALS, and tauopathy (39–45).
NLRs. LRR-containing NLRs form a family of cytosolic PRRs composed of a pyrin domain that receives activation signals, an adaptor protein, and a caspase that is recruited after activation (46). The most relevant member in neurodegeneration is the NLRP3 inflammasome (Figure 1B), which includes the adaptor protein apoptosis-associated speck-like protein containing a caspase recruitment domain (ASC) and caspase-1 (42, 47). Canonical NLRP3 inflammasome activation is a multistep process. It begins with priming through TLR signaling, which increases transcription of NLRP3, pro-IL-1β, pro-IL-18, and the pore-forming protein Gasdermin D (GSDMD) (48, 49). A second signal, usually internalized PAMPs or DAMPs, activates the inflammasome and promotes maturation of caspase-1. Activated caspase-1 then cleaves pro-IL-1β, pro-IL-18, and GSDMD into their active forms for release (50).
Inflammasome activation in connection with AD was first described in 2008 by Halle et al. (51). Aβ fibrils induced IL-1β production by primary microglia in vitro, and NLRP3, caspase-1, and IL-1β were required to recruit microglia to areas of exogenous Aβ deposition (51). Subsequent studies showed that Aβ oligomers and fibrils, but not monomers, interact directly with NLRP3 to activate the inflammasome in a cell-free system and induce IL-1β release in culture (52). Stimulation of primary microglia ex vivo with Aβ oligomers and protofibrils did not impair cell viability, indicating that inflammasome activation in microglia does not result in pyroptosis, which is a common outcome of inflammasome activation (53). In humans with AD, cleaved caspase-1 is increased in tissue from the frontal cortex and hippocampus compared with control tissue (54). These findings were reproduced in APP/PS1 mice, and in that model, NLRP3 deficiency rescued memory impairment and reduced Aβ burden by increasing microglial Aβ clearance (54).
NLRP3 is also highly activated in microglia located in the substantia nigra of postmortem PD patient brains (55). Patients with PD exhibit elevated circulating IL-1β and caspase-1 in the blood and cerebrospinal fluid (CSF) (55, 56). Similar findings were observed in the preformed fibril and 6-OHDA mouse models of PD, in which fibrillar α-syn activated NLRP3, and oral administration of the NLRP3 inhibitor MCC950 reduced motor deficits, neuronal loss, and α-syn accumulation (55). Tau has also been shown to induce NLRP3 inflammasome activation in microglia (57–59). However, the role of inflammasome activity in tauopathy remains unclear, since conflicting results have been reported depending on the mouse model used. For example, loss of NLRP3 or ASC in the PS19 and Thy Tau22 models improved behavioral outcomes and reduced tau aggregation (57, 58, 60), whereas genetic deletion of Nlrp3 did not alter disease progression or outcome in the P301S model (61).
RAGE. The receptor for advanced glycation end products (RAGE) is a multiligand cell surface receptor (Figure 1B). As a transmembrane protein, it contains an extracellular region that binds ligand, a membrane-anchoring region, and a cytosolic region responsible for intracellular signaling (62). RAGE exists in 2 forms; the membrane-bound form contains the full protein structure, while a soluble form contains only the extracellular domain and can neutralize circulating ligands to reduce cellular activation (63). Unlike the other PRR families described above, RAGE primarily recognizes DAMPs. These ligands include AGE, high mobility group box 1 (HMGB1), heat shock proteins (HSPs), S100 proteins, DNA, and in the setting of neurodegenerative disease, aberrant proteins (63). RAGE not only recognizes a wide range of ligands but also initiates diverse intracellular signaling pathways (64).
Studies in AD (65–67), ALS (68, 69), and PD (70–73) have consistently reported elevated RAGE expression on neurons, microglia, and astrocytes, suggesting a broad role for this receptor in neurodegenerative pathology. Functional studies reinforce this idea. Overexpression of RAGE in AD mouse models increased NF-κB translocation and accelerated cognitive impairment (74, 75), whereas RAGE deficiency mitigated amyloid pathology and reduced memory deficits (76). RAGE also contributes to Aβ dynamics at the BBB, as circulating Aβ can bind endothelial RAGE and be transported into the parenchyma, promoting both Aβ accumulation and production of proinflammatory cytokines (77). Consistent with this mechanism, inhibition of RAGE reduced Aβ levels in the Tg2576 model of AD, and an orally administered vaccine containing a RAGE and Aβ complex improved outcomes in APP/PS1 mice by inducing antibodies against both targets (78). Beyond Aβ, RAGE is activated by α-syn and induces production of TNF-α, IL-6, and IL-1β by microglia in culture (79), and RAGE deficiency is similarly beneficial in the SOD1 model of ALS, reducing gliosis and inflammatory marker expression (80). Collectively, these findings demonstrate that RAGE-mediated detection of aberrant proteins amplifies neuroinflammation across multiple neurodegenerative diseases, and a more complete understanding of the pathways downstream of RAGE may help identify therapeutic targets (81).
High mobility group box 1 (HMGB1) is a ubiquitously expressed protein that interacts with DNA to facilitate chromatin binding (82, 83). HMGB1 is detected by TLR2, TLR4, TLR9, and RAGE (84), and it is released by damaged neurons in culture where it promotes microglial activation (Figure 1B) (85, 86). In rats injected intrahippocampally with Aβ, HMGB1 accumulated around dying neurons associated with Aβ plaques. Coinjection of Aβ with exogenous HMGB1 enhanced neuronal loss and impaired microglial Aβ clearance (87). In a rat model of PD induced by 6-OHDA administration, treatment with a mAb targeting HMGB1 ameliorated motor deficits and reduced expression of IL-1β and IL-6 (88). Similarly, anti-HMGB1 mAb treatment delayed loss of grip strength in the SOD1 mouse model of ALS, although this benefit was observed only when treatment began before symptom onset (89).
DNA sensors. Cytosolic DNA sensors (CDSs) represent a class of receptors that detect pathogens or cellular damage. The recently identified cyclic GMP–AMP synthase (cGAS) detects cytosolic double-stranded DNA (dsDNA) and induces a type I interferon (IFN-I) response (Figure 1B) (90). Upon binding dsDNA, cGAS undergoes a conformational change and synthesizes cyclic GMP–AMP (cGAMP) from ATP and GTP (91). cGAMP then binds stimulator of IFN genes (STING), triggering phosphorylation and nuclear translocation of IFN regulatory factor 3 (IRF3) and induction of IFN-I expression (92, 93).
Unlike TLR2 and TLR4, TLR9 resides within late endosomes and detects intracellular DNA containing unmethylated cytosine–guanine (CpG) motifs (94). TLR9 activity is commonly assessed using type B CpG oligodeoxynucleotides (ODNs) (95–97). Activation of TLR9 was neuroprotective in the APP, TgSwDI, and 3xTg AD mouse models by enhancing immune responses and decreasing Aβ burden, although it also promoted tau phosphorylation (94–97). These findings highlight that DNA sensing within the endolysosomal system can modulate AD-related pathology and have prompted investigation into how cytosolic DNA sensing pathways contribute more broadly to neurodegeneration.
Among these mechanisms, the cGAS–STING pathway has emerged as a central modulator of neuroinflammation across multiple neurological disorders as reviewed extensively elsewhere (98–100). In the 5xFAD model, microglia interacting with amyloid plaques containing nucleic acids activated an IFN-1 signature that exacerbated Aβ pathology (101). Similar upregulation of IFN-stimulated genes (ISGs), consistent with activation of cytosolic DNA sensing, occurred in spinal cord astrocytes in the SOD1 model of ALS (102), demonstrating that these pathways are engaged across neurodegenerative contexts. This interpretation is further supported by detection of cGAS in human AD brain tissue, 5xFAD mice, P301S tauopathy mice, and AppNL-G-F/hTau double-knock-in mice (103–105). In these animal models, genetic ablation of cGAS or pharmacologic inhibition of STING reduced Aβ deposition, limited immune activation, and improved cognitive outcomes. Immunostaining revealed colocalization of STING with IRF3 in CD68-expressing cells, implicating microglial cGAS–STING signaling as a major contributor to disease-associated inflammation (103). Similar mechanisms extend to other proteinopathies: α-syn preformed fibrils induced DNA damage and activation of cGAS–STING in mixed glial cultures and in striatal microglia in a corresponding PD model, and loss of STING attenuated the IFN response and conferred neuroprotection (106).
Mitochondrial DNA (mtDNA) plays a pivotal role in activating cGAS–STING signaling during neurodegeneration. Under oxidative stress, mitochondria release ATP, ROS, and mtDNA into the cytosol (107), where the latter can engage cGAS (Figure 1B) (108). In P301S tauopathy mice, pathogenic tau triggered cytosolic release of mtDNA from microglia, initiating cGAS–STING signaling and upregulation of IFN-I and ISGs, whereas cGAS deficiency restored cognition and prevented neuronal loss (104). TDP-43 behaved similarly by entering mitochondria of iPSC-derived motor neurons and promoting mtDNA release, leading to cGAS–STING activation. Deleting or inhibiting STING in TDP-43–overexpressing mice prevented IFN-I induction and cortical degeneration (109). Beyond these models, heightened cGAS–STING signaling was also detected in mice carrying the 2 strongest genetic risk factors for AD, the APOE ε4 allele and the TREM2 R47H variant, and treatment with a cGAS inhibitor ameliorated tau-related pathology in that model (110). In parallel, recent work demonstrates that Aβ-induced oxidative stress drives cytosolic release of oxidized mtDNA fragments that adopt a Z conformation, which are sensed by Z-DNA–binding protein 1 (ZBP1) to activate RIPK1 and promote proinflammatory gene transcription in an AD mouse model (111). Collectively, these studies establish that activation of DNA-sensing pathways, whether endosomal or cytosolic, substantially contributes to neuroinflammation and neurodegenerative progression, providing a strong mechanistic rationale for therapeutic strategies targeting these innate immune axes.
TREM2 signaling. Microglia are the sentinels and first responders of the CNS parenchyma, and microgliosis, particularly in regions with aberrant protein accumulation, is a well-established hallmark of most neurodegenerative diseases (1, 112–115). A unique feature of microglia is their high expression of the triggering receptor expressed on myeloid cells 2 (TREM2) (Figure 1B). Early genetic studies in patients with AD identified the R47H and R62H SNP variants of TREM2 as risk factors at a magnitude similar to that associated with possession of one APOE ε4 allele (116, 117). These discoveries positioned microglia as central players in neurodegenerative disease pathogenesis and accelerated efforts to clarify their functional roles. As research tools have advanced, our understanding of microglial heterogeneity and stimulus-dependent activation states has grown rapidly. A major conceptual advance in this area was the identification of disease-associated microglia (DAMs) in AD using single-cell sequencing approaches (118). During the transition from homeostatic to reactive states, microglia first downregulate markers such as P2RY12, CX3CR1, and TMEM119 (119), and then, in a TREM2-dependent manner, upregulate gene signatures associated with antigen presentation, lipid metabolism, and phagocytosis (120).
Mechanistic studies of TREM2 signaling demonstrate that this receptor activates a complex intracellular cascade that shapes microglial responsiveness during neurodegeneration. Although the R47H and R62H variants have been proposed to impair TREM2 function, animal studies manipulating TREM2 expression have produced mixed results (121, 122). Downstream of TREM2, activation of spleen tyrosine kinase (Syk) and engagement of the PI3K/AKT/GSK3β/mTOR pathway support microglial metabolic programs and phagocytic capacity (123). Reflecting the importance of this signaling axis, microglial Syk knockout in 5xFAD mice impaired Aβ uptake, increased Aβ burden, and worsened memory deficits by disrupting DAM-associated transcriptional and metabolic profiles (123, 124). Conversely, overexpression of human TREM2 in 5xFAD mice enhanced DAM gene expression, improved microglial phagocytic activity, and produced a protective phenotype (125). In line with these findings, TREM2 deficiency in 5xFAD mice increased Aβ burden, suppressed microglial activation, and exacerbated disease progression (126, 127).
Despite this overall pattern, the consequences of TREM2 loss vary across disease stage and neurodegenerative model. In the APP/PS1 model of AD, TREM2 deficiency delayed early amyloid plaque accumulation but ultimately exacerbated pathology at later stages (128, 129). Loss of TREM2 also impaired microglial clustering around plaques, a finding mirrored in postmortem AD brain tissue from individuals carrying R47H and R62H haplodeficient variants (128–130). Beyond AD, TREM2 signaling appears neuroprotective in mice expressing human TDP-43, in which TREM2-deficient microglia failed to phagocytose TDP-43 inclusions and remained in a homeostatic state (131). In tauopathy models, the effects are more complex. In the PS19 mouse line, TREM2 deficiency reduced microgliosis and attenuated microglial activation without altering pTau aggregation (132). However, a study using P301S mice argued that full Trem2 knockout produces an artificially protective phenotype and that Trem2 haplosufficiency, which more accurately reflects human haplodeficiency, is detrimental (133). Collectively, these studies establish TREM2 signaling as a central regulator of microglial activation and function across multiple neurodegenerative diseases and highlight the therapeutic potential of modulating DAM states (134).
MHCII presentation. As myeloid-derived immune cells, microglia also function as antigen-presenting cells capable of processing antigens and presenting them to T cells through MHCII (135). Immunostaining of the CNS in TE4 tauopathy mice carrying human APOE4 showed microglia with elevated MHCII expression concentrated in tau-rich regions. Moreover, DAM-like CD11c+ microglia were frequently positioned adjacent to CD8+ T cells, consistent with potential cellular interactions. Similar patterns were observed in the parenchyma of APP/PS1 mice, where MHCII+ microglia were present near amyloid plaques, although they appeared more sparsely distributed (136). In the P301S model of tauopathy, a substantial population of CD11c+ MHCII+ microglia emerged in the spinal cord of symptomatic mice (137). MHCII expression is likewise increased in microglia in response to α-syn overexpression in vivo. When primary microglia were cultured with α-syn ex vivo, antigen processing was enhanced, and the microglia were able to induce activation and proliferation of CD4+ T cells. MHCII deficiency reduced microgliosis and prevented α-syn–induced neuronal death (138). Collectively, these studies demonstrate extensive coordination between microglia and the adaptive immune response in neurodegenerative disease and emphasize the importance of antigen presentation pathways in disease progression.
-
T cell responses
Adaptive immune involvement has emerged as a shared feature across several neurodegenerative diseases. CD4+ and CD8+ T cells were first observed in the brains of patients with AD in the 1980s (Figure 2) (139, 140). Ongoing work continues to define T cell subpopulations and clarify how the adaptive immune system contributes to neurodegenerative diseases. Overall, these early discoveries established T cells as important immune constituents within the degenerating CNS, prompting deeper investigation into their roles.
 Figure 2
Figure 2CNS distribution of CD4+ and CD8+ T cells across neurodegenerative diseases. Brain regions where CD8+ and CD4+ T cells have been detected in neurodegenerative diseases. MTG, middle temporal gyrus; SN, substantia nigra; VH, ventral horn; Thelp, helper; Teff, effector.
CD4+ T cells. Growing evidence indicates that CD4+ T cells exhibit diverse and disease-specific functions across multiple neurodegenerative disorders (Figure 2). In the CSF of patients with AD, the frequency of CD4+ T cells decreases over time, yet these cells show evidence of clonal expansion with reduced diversity (141). Transfusion of Aβ-restricted CD4+ T cells into APP/PS1 transgenic mice was neuroprotective and prevented cognitive decline while showing little infiltration into the CNS (142). Likewise, transfer of Aβ-specific CD4+ T cells polarized toward a T helper 2 (Th2) phenotype into APP/PS1 mice reversed cognitive decline and synaptic loss (143). CD4+ T cells are also recruited into brain parenchyma in ALS, PD, and LBD (144–148). In the SOD1 ALS model, loss of CD4+ T cells in RAG2 or TCRβ-deficient mice reduced survival and diminished inflammatory gene expression in brain and spinal cord parenchyma (149, 150). In contrast, α-syn–specific CD4+ T cells in PD models were neurotoxic and promoted inflammation in Rag1-knockout mice (144, 147). Overexpression of α-syn enhanced recruitment of IFN-γ–producing CD4+ T cells, and depletion of these cells reduced neuronal loss (151). In LBD, CXCR4-expressing CD4+ T cells and the CXCR4 ligand CXCL12 were increased in the CSF, and CXCL12 levels correlated with neuroaxonal damage (145). Collectively, these studies indicate that CD4+ T cells can be protective in some neurodegenerative contexts but pathogenic in others, particularly in the presence of α-syn.
The inconsistencies among diseases may reflect differences in CD4+ T cell subpopulations. Tregs, defined by expression of FOXP3, play essential roles in immune regulation (152). Total (CD25 high and CD127 low to negative) and resting (CD45RA+ and CD25 dim) Tregs were reduced in peripheral blood from patients with AD (153). In ALS, the ratio of Tregs to total CD4+ T cells inversely correlated with disease progression (146). Further analysis showed that high frequencies of CD4+FOXP3neg effector T cells in blood and CSF predicted poor survival, whereas high levels of activated Tregs and a high ratio of activated-to-resting Tregs were associated with improved survival (154). In PD, increased CD45RO+FAS+ effector memory CD4+ T cells were observed along with dysfunctional Tregs (155). Patients with mutations in the huntingtin (HTT) gene showed reduced Treg frequencies and increased IL-17–expressing Th17 cells in CSF. Whether enrichment of beneficial CD4+ T cell subsets could slow disease remains an open question. In one study, PD-1 blockade increased the abundance of FOXP3+CD4+ Tregs, including PD-1+FOXP3+CD4+ subsets, in TE4 tauopathy mice, which was associated with reduced pTau accumulation and neurodegeneration (136). These observations together suggest that the balance between regulatory and effector CD4+ T cell subsets may critically shape neurodegenerative disease progression and could represent a therapeutic target.
CD8+ T cells. As interest in adaptive immunity in neurodegenerative disease has grown, CD8+ T cells have become increasingly recognized as important contributors to CNS pathology, but their disease context–dependent functions remain unclear (Figure 2). Extravascular CD3+ and CD8+ T cells were detected in the hippocampus of postmortem AD brains, and CD3+ T cell numbers correlated with tau pathology but not amyloid plaques (156, 157). Depletion of CD8+ T cells in APP/PS1 mice did not alter disease course, though it did change neuronal gene expression. Notably, depletion in this study was initiated at 12 months of age when marked pathology was already present (157). In PD, CD8+ T cell numbers increased and correlated with neuronal death, although it remains uncertain whether this is causative or reactive (158). α-syn–specific T cells emerge in the blood before motor symptoms in PD, peak soon after symptom onset, and decline as disease progresses. Reconstitution experiments in RAG-deficient mice demonstrated that both CD4+ and CD8+ T cells can independently induce neuronal loss in mice overexpressing human A53T α-syn through antigen-specific responses (144). CD8+ T cells were also present in patients with FTD with the P301L mutation and in the hippocampus of THY Tau22 mice. In that model, depleting CD4+ and CD8+ T cells with an anti-CD3 antibody rescued behavior defects despite no effect on pTau deposition (159). In TE4 tauopathy mice, microglia facilitated recruitment of T cells to the dentate gyrus, and depletion of microglia or CD4+ and CD8+ T cells reduced tau pathology and neurological decline (136). Overall, the evidence points to highly context-dependent roles for CD8+ T cells in neurodegenerative disease.
To understand these divergent effects, attention has shifted toward defining CD8+ T cell subsets and the mechanisms directing their localization and activity in the CNS. CD8+ tissue–resident memory (Trm) cells depend on CXCR6 for localization to peripheral tissues (160). In aged APP/PS1 mice, CXCR6+ CD8+ T cells were enriched in the hippocampus relative to blood, and their transcriptomic profile resembled antiviral and antitumor responses (161). In 5xFAD mice, neuroprotective CD8+ T cells accumulated in the brain and restricted Aβ plaque growth. Single-cell sequencing identified CXCL16 expression by microglia and CXCR6 expression by CD8+ T cells, and this signaling axis was required for their protective effect. Combined single-cell and TCR sequencing revealed clonally expanded CD4+ and CD8+ T cells that transitioned from activated to exhausted states, with the activated subset exhibiting neuroprotective properties (162). This axis appears conserved in humans, as cognitively impaired individuals showed elevated CXCR6+CD8+ T effector memory (Tem) cells in CSF. Immunostaining of AD brain tissue confirmed CXCR6 expression on CD3+ T cells and CXCL16 expression by Iba1+ microglia, supporting microglia-mediated recruitment into the parenchyma (163). Extending these findings to the leptomeningeal compartment, a recent study identified clonally expanded PD-1+ CD8+ Trm cells in the leptomeninges of patients with AD and ALS, with substantial overlap between brain and leptomeningeal TCR repertoires, while additional tissue-specific clones emerged in AD. In AD, the degree of CD8+ Trm clonal expansion correlated positively with microglial TGFB2 expression, raising the possibility of functional crosstalk between meningeal T cells and parenchymal immune cells (164). Together, these findings underscore the importance of Trm-like CD8+ T cell populations in shaping immune dynamics during neurodegeneration, while raising important questions regarding how distinct memory CD8+ T cell subsets contribute to disease progression.
Additional memory CD8+ T cell subsets with strong cytotoxic potential include terminally differentiated effector memory (Temra) cells (165). Temra cells accumulated in the CSF during mild cognitive impairment, an early stage of AD (166). In another study, increased numbers of CD8+ Temra and Tem cells in the hippocampus and leptomeninges correlated with cognitive decline in AD. Temra cells also clonally expand in the CSF of patients with AD, with some clones recognizing EBV, although this does not imply a causal relationship between EBV exposure and AD (167). A sex-specific increase in CD8+ Tem and Temra cells has also been reported in females with idiopathic PD who exhibit reduced CD8+ Tregs and an elevated Temra-to-Treg ratio (168). In ALS4, caused by mutations in senataxin (SETX), patients show clonally expanded CD8+ Temra cells in circulation and CD8+ T cells in the ventral horn of the spinal cord. Mice carrying the L398S Setx mutation similarly display expanded PD-1+CD8+ Temra cells across spinal cord, brain, and blood, indicating that antigen-specific CD8+ T cell responses contribute to disease development (169). These findings highlight the diverse ways in which memory CD8+ T cell subsets become activated and expanded across neurodegenerative diseases, paving the way for the identification of additional populations with specialized functions.
As research tools advance, T cell subsets continue to be defined with increasing precision. A recently described population of granzyme K–expressing (GzmK-expressing) CD8+ T cells has been implicated in chronic tissue inflammation and complement activation (170–174). Although less cytolytic than other granzymes, GzmK promotes antiviral responses through inflammatory mechanisms (173, 175). GzmK+ CD8+ T cells were identified in the CSF of patients with ALS, in post mortem brains of individuals with ALS, AD, chronic traumatic encephalopathy (CTE), and advanced age, and in the spinal cords of P301S tauopathy mice (137, 176, 177). In sporadic ALS, these cells clonally expanded in CNS and blood and were enriched in parenchyma and perivascular regions compared with controls (177). In the P301S model, microglia upregulated CD11c and MHCII, supporting pTau clearance. Under conditions of sustained inflammation, clonally expanded PD-1+CD103+CD8+ Trm cells with a noneffector transcriptional profile infiltrated the spinal cord parenchyma, interacted with maladaptive microglia, and deposited extracellular GzmK. CD8 deficiency exacerbated pTau accumulation and produced a distressed microglial population containing neuronal transcripts, leading to accelerated neurological decline (137). These findings suggest that GzmK+CD8+ T cells are protective in this tauopathy model. By contrast, in 3xTg AD mice, CD103– CD8+ Trm cells expressed high levels of GzmK and adopted a gene signature associated with neurodegeneration. Depletion of peripheral CD8+ T cells reduced Aβ and pTau accumulation, improving cognition. Coculture experiments confirmed that these CD103–CD8+ Trm cells were directly neurotoxic through GzmK-mediated engagement of protease activation receptor 1 (176). Further work is needed to determine the disease specificity of GzmK+CD8+ T cell functions and whether these cells can be therapeutically targeted.
Overall, CD8+ T cells display diverse roles in neurodegeneration, shaped by tissue residency, antigen specificity, and interactions with microglia. Across diseases, these cells infiltrate affected CNS regions, clonally expand, and adopt diverse transcriptional states ranging from protective to pathogenic, as illustrated by CXCR6+ Trm-like cells, cytotoxic Tem/Temra subsets, and GzmK+ populations. These observations highlight the need to define the drivers of CD8+ T cell activation, the antigens that shape their clonal repertoires, and the microenvironmental factors that determine whether they support repair or promote neurodegeneration. Integrating refined T cell subset classification with mechanistic studies of T cell–microglia interactions will be crucial for developing precise immunomodulatory therapies tailored to specific neurodegenerative contexts.
-
Factors shaping neurodegenerative immune responses
Although our understanding of immunopathogenesis in neurodegenerative disease continues to expand, these disorders remain highly complex, with multifactorial and often unclear etiologies. Insights into the roles of resident and infiltrating immune cells highlight the need to consider additional influences that shape how immune responses affect neuronal health and overall disease risk. Examining aging, repetitive injury, and viral infection provides a framework for understanding how diverse biological and environmental pressures modulate CNS immunity during neurodegeneration (Figure 3).
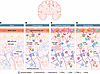 Figure 3
Figure 3External factors that reshape CNS immunity and influence neurodegenerative risk. External factors influence both the initiation and progression of neurodegenerative disease by activating distinct immune pathways. The illustration depicts 4 conditions that alter CNS immunity. (A) In the naive state, intact vasculature, BBB-associated astrocytes, and homeostatic glia maintain neural function. (B) Inflammaging is characterized by BBB decline, infiltration of peripheral immune cells, including granzyme K–expressing CD8+ T cells, and accumulation of circulating material such as fibrinogen, albumin, and ions that drive chronic low-grade inflammation and aberrant protein deposition. (C) Repetitive head injury induces persistent BBB disruption, reduced vascular integrity, and reactive astrocytes and microglia that cluster around accumulating Aβ and pTau; CD8+ T cells deposit granzyme K onto these microglial aggregates. (D) During viral infection, microglia and astrocytes respond to pathogen-associated signals and recruit myelomonocytic cells along with CD4+ and CD8+effector T cells, which utilize IFN-γ, TNF-α, and granzyme A/B/C to control infection.
Inflammaging. Aging is one of the primary risk factors for neurodegenerative diseases such as AD and PD. In youth, homeostatic microglia survey the CNS, astrocytes maintain the BBB, and only limited numbers of T cells enter the parenchyma. With advanced age, however, chronic low-grade inflammation develops in a process termed inflammaging (Figure 3B), during which many of the immune pathways described above become dysregulated (178). For example, plasma mtDNA levels increase after age 50 and correlate with elevated proinflammatory cytokines such as TNF-α and IL-6 (179). Consistently, activation of cGAS in aged mice produced cognitive impairment, neuronal loss, and neuroinflammation similar to that seen in neurodegenerative models (180). Systemic inflammation increases with age, and these immune alterations are accompanied by profound structural and functional changes within the CNS that further heighten vulnerability to neurodegenerative pathology.
Aging also compromises the integrity of the BBB. Increased permeability promotes leukocyte extravasation into the parenchyma (181) and this process is exacerbated by age-related declines in microvasculature density (182, 183). As a result, cytotoxic CD8+ T cells can accumulate and damage neurons through granzyme B activity (184). In humans, a distinct exhausted-like subset of granzyme K-expressing CD8+ T cells expands with age, and aged mice exhibit a PD1+GzmK+CD8+ population that mirrors the phenotype seen in tauopathy models (137, 172, 185). Together, these findings suggest that age-related immune changes can both impair CNS homeostasis and predispose individuals to neurodegenerative disease development.
Repetitive head injury. External insults such as repetitive head injury can further disrupt CNS immune homeostasis and accelerate neurodegenerative processes (Figure 3C). Traumatic brain injury (TBI) is strongly linked to later development of neurodegenerative diseases including AD, PD, and CTE (186–188), even when injuries occur during childhood (189). Following mild TBI, distressed cells release DAMPs such as mtDNA and HMGB1 (190), triggering microglial activation. Microglia clear debris and reinforce weakened regions of the BBB and glia limitans superficialis, which becomes vulnerable following astrocyte death (191–194). However, repeated injuries can cause these barrier-supporting microglia to become reactive and die, leaving openings that allow entry of myelomonocytic cells (195, 196). If the CNS cannot restore homeostasis after injury, risk of CTE increases. CTE is characterized by pTau accumulation and widespread glial activation. Postmortem CTE brains often show additional proteinopathies including TDP-43 inclusions, Aβ plaques, and α-syn deposits (197–200). Across experimental TBI paradigms, injury precipitates acute and chronic proteinopathy, including elevations in total and oligomeric tau (201), transient increases in soluble and insoluble Aβ species (202), enhanced hippocampal Aβ deposition at later time points (203, 204), and broader neurodegenerative protein signatures following repetitive injury (205). Because CTE can only be diagnosed histologically, early detection after TBI is a critical unmet need. Understanding how repeated injuries promote chronic immune activation may reveal mechanisms shared with other neurodegenerative diseases and highlight intervention points independent of age or genetic risk.
CNS viral infection. Like repetitive injury, viral infections can profoundly alter CNS immunity and shape susceptibility to neurodegeneration (Figure 3D). Many immune pathways implicated in neurodegeneration originally evolved to combat invading pathogens, and recent studies link both neurotropic and nonneurotropic viral infections to increased risk for, or accelerated progression of, neurodegenerative diseases (206–210). Viruses access the CNS through several mechanisms, including infection of endothelial cells or astrocytes (211), or through inflammation-induced barrier disruption similar to that seen in aging and TBI (212, 213). Once present, viral products activate microglia and infiltrating myeloid cells through both PAMP- and DAMP-dependent pathways, promoting secretion of IFN-I and other inflammatory mediators involved in viral clearance (214). Consistent with a potential role for herpesviruses in dementia risk, natural experiment–based studies have reported that herpes zoster vaccination (both live-attenuated and recombinant) is associated with reduced incidence of dementia and prolonged dementia diagnosis–free survival (215–217). Although causality and mechanisms remain to be established, these findings suggest that antiviral vaccination or immune modulation may influence dementia risk and disease trajectory.
As the antiviral response progresses, these innate signaling events rapidly recruit adaptive immune cells into the CNS. During early infection, CD4+ and CD8+ T cells traffic into the CNS, with CD8+ T cells comprising the majority of infiltrating lymphocytes and relying on IFN-γ, TNF-α, and perforin for antiviral effector activity (218–220). Repeated peripheral viral infections have been shown to expand and diversify the pool of CD8+ Trm cells in the CNS, thereby enhancing protection against subsequent infections (221, 222). However, persistence of CD8+ T cells after viral clearance can promote chronic microglial activation, synaptic loss, and cognitive deficits (223). Viral products may also interact with pathogenic proteins implicated in neurodegenerative diseases. HSV-1 DNA colocalized with Aβ plaques in both AD and control brains, although unbound HSV-1 DNA was more prevalent in controls, suggesting more efficient viral clearance under normal conditions (224). Aβ has been proposed to function as an antimicrobial peptide and may be upregulated as part of a protective innate response. In 5xFAD mice infected with HSV-1, Aβ plaque formation accelerated and survival improved (225), although other studies found no protective effect across HSV-1 strains (226). These findings support a model in which viral infections, even after resolution, reshape the CNS immune environment in ways that may increase susceptibility to future neurodegenerative processes, while also raising the possibility that certain infections or antiviral responses could transiently modulate pathology in protective or compensatory ways.
-
Concluding remarks
The CNS parenchyma can mount immune responses, yet these responses are carefully regulated to preserve neural function. Microglial engagement of self-derived DAMPs, together with the recruitment of diverse T cell populations, illustrates how multiple immune compartments respond dynamically to pathogenic protein accumulation. Advances such as single-cell sequencing have expanded our understanding of these processes by revealing distinct immune subsets and clarifying how innate and adaptive pathways intersect. Together, these insights establish immune dysregulation as a unifying theme across the spectrum of neurodegenerative disorders.
Neurodegenerative diseases arise from a complex interplay of biological and environmental factors that shape not only the timing and magnitude of immune activation but also the programming of microglia, T cells, and other leukocytes. Aging, repetitive head injury, and viral infection each restructure the CNS immune environment by promoting chronic inflammation, altering microglial and T cell phenotypes, and shifting the balance between protective and damaging responses. Many of the immune pathways involved show context-specific and stage-dependent functions. Microglial activation may aid early clearance of aberrant proteins but become maladaptive with prolonged stimulation, and CD8+ T cells can either support tissue repair or promote neuronal injury depending on antigen specificity, effector programming, and local signaling cues. Appreciating both the temporal dynamics and cell-intrinsic states of immune populations is essential for interpreting immune signatures and for identifying when immunomodulatory intervention is most likely to be beneficial.
These discoveries have strengthened the rationale for developing targeted immunotherapies to slow or prevent neurodegenerative disease progression. Approaches under investigation include modulation of microglial receptors such as TREM2, inhibition of pathways such as cGAS and STING, and strategies that reshape T cell activity through immune checkpoint molecules like PD-1 or by enhancing regulatory T cell function. In the future, adoptive immunotherapies that involve transferring expanded or engineered T cell subsets with neuroprotective properties may offer new opportunities to reinforce beneficial immune programs or counter harmful ones. Continued integration of human and animal studies, high dimensional profiling, and longitudinal sampling will be vital for determining causality, identifying therapeutic windows, and distinguishing adaptive from maladaptive immune remodeling. A deeper understanding of how immune signaling networks interact with genetic, environmental, and age-related stressors will be essential for guiding the development of effective immunomodulatory therapies.
-
Conflict of interest
The authors have declared that no conflict of interest exists.
-
Funding support
This work is the result of NIH funding, in whole or in part, and is subject to the NIH Public Access Policy. Through acceptance of this federal funding, the NIH has been given a right to make the work publicly available in PubMed Central. The contributions of the NIH authors are considered Works of the United States Government. The findings and conclusions presented in this paper are those of the authors and do not necessarily reflect the views of the NIH or the U.S. Department of Health and Human Services.
- The Defense Health Agency (CA#HU00012520039/HJF#10002).
- The Intramural Research Program of the National Institute of Neurological Disorders & Stroke (NINDS) within the National Institutes of Health (NIH).
-
Acknowledgments
We thank Ethan Tyler in the NIH Medical Arts Design Section for his help with generating all the figure illustrations.
Address correspondence to: Dorian B. McGavern, National Institutes of Health, Building 10, Room 5N240C, Bethesda, Maryland 20892, USA. Phone: 301.443.7949; Email: mcgavernd@mail.nih.gov.
-
Footnotes
Copyright: © 2026, Latour et al. This is an open access article published under the terms of the Creative Commons Attribution 4.0 International License.
Reference information: J Clin Invest. 2026;136(8):e199850. https://doi.org/10.1172/JCI199850.
-
References
- Wilson DM, et al. Hallmarks of neurodegenerative diseases. Cell. 2023;186(4):693–714.
- Heneka MT, et al. Neuroinflammation in Alzheimer disease. Nat Rev Immunol. 2025;25(5):321–352.
- Johnson AM, Lukens JR. Emerging roles for innate and adaptive immunity in tauopathies. Cell Rep. 2025;44(9):116232.
- Mason HD, McGavern DB. How the immune system shapes neurodegenerative diseases. Trends Neurosci. 2022;45(10):733–748.
- Chen X, Holtzman DM. Emerging roles of innate and adaptive immunity in Alzheimer’s disease. Immunity. 2022;55(12):2236–2254.
- Pellicano M, et al. Immune profiling of Alzheimer patients. J Neuroimmunol. 2012;242(1-2):52–59.
- Mohan M, et al. Parkinson’s disease: exploring the systemic immune mechanisms through molecular investigations. Inflammopharmacology. 2025;33(7):3679–3699.
- Terrabuio E, et al. The role of the CD8+ T cell compartment in ageing and neurodegenerative disorders. Front Immunol. 2023;14:1233870.
- Janeway CA, JrInnate immune recognition. Annu Rev Immunol. 2002;20:197–216.
- Matzinger P. The danger model: a renewed sense of self. Science. 2002;296(5566):301–305.
- Kumar H, et al. Pathogen recognition by the innate immune system. Int Rev Immunol. 2011;30(1):16–34.
- Bsibsi M, et al. Broad expression of Toll-like receptors in the human central nervous system. J Neuropathol Exp Neurol. 2002;61(11):1013–1021.
- Cameron JS, et al. Toll-like receptor 3 is a potent negative regulator of axonal growth in mammals. J Neurosci. 2007;27(47):13033–13041.
- Owens T. Toll-like receptors in neurodegeneration. Curr Top Microbiol Immunol. 2009;336:105–120.View this article via: PubMed Google Scholar
- Tang SC, et al. Pivotal role for neuronal Toll-like receptors in ischemic brain injury and functional deficits. Proc Natl Acad Sci U S A. 2007;104(34):13798–13803.
- Tang SC, et al. Toll-like receptor-4 mediates neuronal apoptosis induced by amyloid beta-peptide and the membrane lipid peroxidation product 4-hydroxynonenal. Exp Neurol. 2008;213(1):114–121.
- Hoffmann JA. The immune response of Drosophila. Nature. 2003;426(6962):33–38.
- Kawai T, Akira S. Toll-like receptors and their crosstalk with other innate receptors in infection and immunity. Immunity. 2011;34(5):637–650.
- Akira S, et al. Pathogen recognition and innate immunity. Cell. 2006;124(4):783–801.
- Beutler BA. TLRs and innate immunity. Blood. 2009;113(7):1399–1407.
- Kumar V. Toll-like receptors in the pathogenesis of neuroinflammation. J Neuroimmunol. 2019;332:16–30.
- Castellanos-Molina A, et al. Constitutive DAMPs in CNS injury: From preclinical insights to clinical perspectives. Brain Behav Immun. 2024;122:583–595.
- Zindel J, Kubes P. DAMPs, PAMPs, and LAMPs in immunity and sterile inflammation. Annu Rev Pathol. 2020;15:493–518.
- Lester E, et al. Tau aggregates are RNA-protein assemblies that mislocalize multiple nuclear speckle components. Neuron. 2021;109(10):1675–1691.
- Kampers T, et al. RNA stimulates aggregation of microtubule-associated protein tau into Alzheimer-like paired helical filaments. FEBS Lett. 1996;399(3):344–349.
- Abskharon R, et al. Cryo-EM structure of RNA-induced tau fibrils reveals a small C-terminal core that may nucleate fibril formation. Proc Natl Acad Sci U S A. 2022;119(15):e2119952119.
- Matsuo K, et al. RNA G-quadruplexes form scaffolds that promote neuropathological α-synuclein aggregation. Cell. 2024;187(24):6835–6848.
- Che MX, et al. TDP-35 sequesters TDP-43 into cytoplasmic inclusions through binding with RNA. FEBS Lett. 2015;589(15):1920–1928.
- Khmeleva SA, et al. Zinc-mediated binding of nucleic acids to amyloid-β aggregates: role of histidine residues. J Alzheimers Dis. 2016;54(2):809–819.
- Rangasamy SB, et al. Selective disruption of TLR2-MyD88 interaction inhibits inflammation and attenuates Alzheimer’s pathology. J Clin Invest. 2018;128(10):4297–4312.
- Liu S, et al. TLR2 is a primary receptor for Alzheimer’s amyloid β peptide to trigger neuroinflammatory activation. J Immunol. 2012;188(3):1098–1107.
- Song M, et al. TLR4 mutation reduces microglial activation, increases Aβ deposits and exacerbates cognitive deficits in a mouse model of Alzheimer’s disease. J Neuroinflammation. 2011;8:92.
- Udan ML, et al. Toll-like receptors 2 and 4 mediate Abeta(1-42) activation of the innate immune response in a human monocytic cell line. J Neurochem. 2008;104(2):524–533.
- Jin JJ, et al. Toll-like receptor 4-dependent upregulation of cytokines in a transgenic mouse model of Alzheimer’s disease. J Neuroinflammation. 2008;5:23.
- Richard KL, et al. Toll-like receptor 2 acts as a natural innate immune receptor to clear amyloid beta 1-42 and delay the cognitive decline in a mouse model of Alzheimer’s disease. J Neurosci. 2008;28(22):5784–5793.
- Zhou C, et al. Genomic deletion of TLR2 induces aggravated white matter damage and deteriorated neurobehavioral functions in mouse models of Alzheimer’s disease. Aging (Albany NY). 2019;11(17):7257–7273.
- Lehnardt S, et al. A vicious cycle involving release of heat shock protein 60 from injured cells and activation of toll-like receptor 4 mediates neurodegeneration in the CNS. J Neurosci. 2008;28(10):2320–2331.
- Leitner GR, et al. Targeting toll-like receptor 4 to modulate neuroinflammation in central nervous system disorders. Expert Opin Ther Targets. 2019;23(10):865–882.
- Hughes CD, et al. Picomolar concentrations of oligomeric alpha-synuclein sensitizes TLR4 to play an initiating role in Parkinson’s disease pathogenesis. Acta Neuropathol. 2019;137(1):103–120.
- Zhao W, et al. Extracellular mutant SOD1 induces microglial-mediated motoneuron injury. Glia. 2010;58(2):231–243.
- Scheiblich H, et al. Microglial NLRP3 inflammasome activation upon TLR2 and TLR5 ligation by distinct α-synuclein assemblies. J Immunol. 2021;207(8):2143–2154.
- Yang J, et al. TLR4 cross-talk with NLRP3 inflammasome and complement signaling pathways in Alzheimer’s Disease. Front Immunol. 2020;11:724.
- Campolo M, et al. TLR4 absence reduces neuroinflammation and inflammasome activation in Parkinson’s diseases in vivo model. Brain Behav Immun. 2019;76:236–247.
- Dutta D, et al. Tau fibrils induce glial inflammation and neuropathology via TLR2 in Alzheimer’s disease-related mouse models. J Clin Invest. 2023;133(18):e161987.
- Kim C, et al. Neuron-released oligomeric α-synuclein is an endogenous agonist of TLR2 for paracrine activation of microglia. Nat Commun. 2013;4:1562.
- Chou WC, et al. The NLR gene family: from discovery to present day. Nat Rev Immunol. 2023;23(10):635–654.
- Hanslik KL, Ulland TK. The role of microglia and the Nlrp3 inflammasome in Alzheimer’s disease. Front Neurol. 2020;11:570711.
- Broz P, et al. The gasdermins, a protein family executing cell death and inflammation. Nat Rev Immunol. 2020;20(3):143–157.
- Weber ANR, et al. Recent insights into the regulatory networks of NLRP3 inflammasome activation. J Cell Sci. 2020;133(23):jcs248344.
- Heneka MT, et al. Inflammasome signalling in brain function and neurodegenerative disease. Nat Rev Neurosci. 2018;19(10):610–621.
- Halle A, et al. The NALP3 inflammasome is involved in the innate immune response to amyloid-beta. Nat Immunol. 2008;9(8):857–865.
- Nakanishi A, et al. Amyloid β directly interacts with NLRP3 to initiate inflammasome activation: identification of an intrinsic NLRP3 ligand in a cell-free system. Inflamm Regen. 2018;38:27.
- Luciunaite A, et al. Soluble Aβ oligomers and protofibrils induce NLRP3 inflammasome activation in microglia. J Neurochem. 2020;155(6):650–661.
- Heneka MT, et al. NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/PS1 mice. Nature. 2013;493(7434):674–678.
- Gordon R, et al. Inflammasome inhibition prevents α-synuclein pathology and dopaminergic neurodegeneration in mice. Sci Transl Med. 2018;10(465):eaah4066.
- Blum-Degen D, et al. Interleukin-1 beta and interleukin-6 are elevated in the cerebrospinal fluid of Alzheimer’s and de novo Parkinson’s disease patients. Neurosci Lett. 1995;202(1-2):17–20.
- Stancu IC, et al. Aggregated Tau activates NLRP3-ASC inflammasome exacerbating exogenously seeded and non-exogenously seeded Tau pathology in vivo. Acta Neuropathol. 2019;137(4):599–617.
- Ising C, et al. NLRP3 inflammasome activation drives tau pathology. Nature. 2019;575(7784):669–673.
- Zhang L, et al. Tau induces inflammasome activation and microgliosis through acetylating NLRP3. Clin Transl Med. 2024;14(3):e1623.
- Jiang S, et al. Proteopathic tau primes and activates interleukin-1β via myeloid-cell-specific MyD88- and NLRP3-ASC-inflammasome pathway. Cell Rep. 2021;36(12):109720.
- Paesmans I, et al. NLRP3 inflammasome activation and pyroptosis are dispensable for tau pathology. Front Aging Neurosci. 2024;16:1459134.
- Fritz G. RAGE: a single receptor fits multiple ligands. Trends Biochem Sci. 2011;36(12):625–632.
- Ding Q, Keller JN. Evaluation of rage isoforms, ligands, and signaling in the brain. Biochim Biophys Acta. 2005;1746(1):18–27.
- Kinscherf NA, Pehar M. Role and therapeutic potential of RAGE signaling in neurodegeneration. Curr Drug Targets. 2022;23(12):1191–1209.
- Miller MC, et al. Hippocampal RAGE immunoreactivity in early and advanced Alzheimer’s disease. Brain Res. 2008;1230:273–280.
- Sasaki N, et al. Immunohistochemical distribution of the receptor for advanced glycation end products in neurons and astrocytes in Alzheimer’s disease. Brain Res. 2001;888(2):256–262.
- Lue LF, et al. Involvement of microglial receptor for advanced glycation endproducts (RAGE) in Alzheimer’s disease: identification of a cellular activation mechanism. Exp Neurol. 2001;171(1):29–45.
- Casula M, et al. Toll-like receptor signaling in amyotrophic lateral sclerosis spinal cord tissue. Neuroscience. 2011;179:233–243.
- Juranek JK, et al. Receptor for advanced glycation end products and its inflammatory ligands are upregulated in amyotrophic lateral sclerosis. Front Cell Neurosci. 2015;9:485.
- Teismann P, et al. Receptor for advanced glycation endproducts (RAGE) deficiency protects against MPTP toxicity. Neurobiol Aging. 2012;33(10):2478–2490.
- Gasparotto J, et al. Targeted inhibition of RAGE in substantia nigra of rats blocks 6-OHDA-induced dopaminergic denervation. Sci Rep. 2017;7(1):8795.
- Guerrero E, et al. Recent advances in α-synuclein functions, advanced glycation, and toxicity: implications for Parkinson’s disease. Mol Neurobiol. 2013;47(2):525–536.
- Jiang X, et al. RAGE and its emerging role in the pathogenesis of Parkinson’s disease. Neurosci Lett. 2018;672:65–69.
- Fang F, et al. RAGE-dependent signaling in microglia contributes to neuroinflammation, Abeta accumulation, and impaired learning/memory in a mouse model of Alzheimer’s disease. FASEB J. 2010;24(4):1043–1055.
- Arancio O, et al. RAGE potentiates Abeta-induced perturbation of neuronal function in transgenic mice. EMBO J. 2004;23(20):4096–4105.
- Fang F, et al. RAGE mediates Aβ accumulation in a mouse model of Alzheimer’s disease via modulation of β- and γ-secretase activity. Hum Mol Genet. 2018;27(6):1002–1014.
- Deane R, et al. RAGE mediates amyloid-beta peptide transport across the blood-brain barrier and accumulation in brain. Nat Med. 2003;9(7):907–913.
- Webster SJ, et al. An aqueous orally active vaccine targeted against a RAGE/AB complex as a novel therapeutic for Alzheimer’s disease. Neuromolecular Med. 2012;14(2):119–130.
- Long H, et al. Interaction of RAGE with α-synuclein fibrils mediates inflammatory response of microglia. Cell Rep. 2022;40(12):111401.
- Lee JD, et al. Absence of receptor for advanced glycation end product (RAGE) reduces inflammation and extends survival in the hSOD1G93A mouse model of amyotrophic lateral sclerosis. Mol Neurobiol. 2020;57(10):4143–4155.
- Koerich S, et al. Receptors for Advanced Glycation End Products (RAGE): Promising Targets Aiming at the Treatment of Neurodegenerative Conditions. Curr Neuropharmacol. 2023;21(2):219–234.
- Goodwin GH, et al. A new group of chromatin-associated proteins with a high content of acidic and basic amino acids. Eur J Biochem. 1973;38(1):14–19.
- Bianchi ME, Beltrame M. Upwardly mobile proteins. Workshop: the role of HMG proteins in chromatin structure, gene expression and neoplasia. EMBO Rep. 2000;1(2):109–114.
- Castro-Gomez S, Heneka MT. Innate immune activation in neurodegenerative diseases. Immunity. 2024;57(4):790–814.
- Kim JB, et al. HMGB1, a novel cytokine-like mediator linking acute neuronal death and delayed neuroinflammation in the postischemic brain. J Neurosci. 2006;26(24):6413–6421.
- Gao HM, et al. HMGB1 acts on microglia Mac1 to mediate chronic neuroinflammation that drives progressive neurodegeneration. J Neurosci. 2011;31(3):1081–1092.
- Takata K, et al. High mobility group box protein-1 inhibits microglial Abeta clearance and enhances Abeta neurotoxicity. J Neurosci Res. 2004;78(6):880–891.
- Sasaki T, et al. Anti-high mobility group box 1 antibody exerts neuroprotection in a rat model of Parkinson’s disease. Exp Neurol. 2016;275 Pt 1:220–231.
- Lee JD, et al. Therapeutic blockade of HMGB1 reduces early motor deficits, but not survival in the SOD1G93A mouse model of amyotrophic lateral sclerosis. J Neuroinflammation. 2019;16(1):45.
- Sun L, et al. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science. 2013;339(6121):786–791.
- Zhang X, et al. The cytosolic DNA sensor cGAS forms an oligomeric complex with DNA and undergoes switch-like conformational changes in the activation loop. Cell Rep. 2014;6(3):421–430.
- Ablasser A, et al. cGAS produces a 2’-5’-linked cyclic dinucleotide second messenger that activates STING. Nature. 2013;498(7454):380–384.
- Tanaka Y, Chen ZJ. STING specifies IRF3 phosphorylation by TBK1 in the cytosolic DNA signaling pathway. Sci Signal. 2012;5(214):ra20.
- Cacace R, et al. Mutated Toll-like receptor 9 increases Alzheimer’s disease risk by compromising innate immunity protection. Mol Psychiatry. 2023;28(12):5380–5389.
- Scholtzova H, et al. Amyloid β and Tau Alzheimer’s disease related pathology is reduced by Toll-like receptor 9 stimulation. Acta Neuropathol Commun. 2014;2:101. View this article via: PubMed Google Scholar
- Scholtzova H, et al. Innate immunity stimulation via toll-like receptor 9 ameliorates vascular amyloid pathology in Tg-SwDI mice with associated cognitive benefits. J Neurosci. 2017;37(4):936–959.
- Scholtzova H, et al. Induction of toll-like receptor 9 signaling as a method for ameliorating Alzheimer’s disease-related pathology. J Neurosci. 2009;29(6):1846–1854.
- Kaur A, Aran KR. Unraveling the cGAS-STING pathway in Alzheimer’s disease: A new Frontier in neuroinflammation and therapeutic strategies. Neuroscience. 2025;573:430–441.
- Zhang Y, et al. The cGAS-STING pathway drives neuroinflammation and neurodegeneration via cellular and molecular mechanisms in neurodegenerative diseases. Neurobiol Dis. 2024;202:106710.
- Moya GE, et al. Evidence for the role of mitochondrial DNA release in the inflammatory response in neurological disorders. Int J Mol Sci. 2021;22(13):7030.
- Roy ER, et al. Type I interferon response drives neuroinflammation and synapse loss in Alzheimer disease. J Clin Invest. 2020;130(4):1912–1930.
- Wang R, et al. Activation of interferon signaling pathways in spinal cord astrocytes from an ALS mouse model. Glia. 2011;59(6):946–958.
- Xie X, et al. Activation of innate immune cGAS-STING pathway contributes to Alzheimer’s pathogenesis in 5×FAD mice. Nat Aging. 2023;3(2):202–212.
- Udeochu JC, et al. Tau activation of microglial cGAS-IFN reduces MEF2C-mediated cognitive resilience. Nat Neurosci. 2023;26(5):737–750.
- Chung S, et al. Blockade of STING activation alleviates microglial dysfunction and a broad spectrum of Alzheimer’s disease pathologies. Exp Mol Med. 2024;56(9):1936–1951.
- Hinkle JT, et al. STING mediates neurodegeneration and neuroinflammation in nigrostriatal α-synucleinopathy. Proc Natl Acad Sci U S A. 2022;119(15):e2118819119.
- Shimada K, et al. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity. 2012;36(3):401–414.
- Quan S, et al. The neuroimmune nexus: unraveling the role of the mtDNA-cGAS-STING signal pathway in Alzheimer’s disease. Mol Neurodegener. 2025;20(1):25.
- Yu CH, et al. TDP-43 triggers mitochondrial DNA release via mPTP to activate cGAS/STING in ALS. Cell. 2020;183(3):636–649.
- Carling GK, et al. Alzheimer’s disease-linked risk alleles elevate microglial cGAS-associated senescence and neurodegeneration in a tauopathy model. Neuron. 2024;112(23):3877–3896.
- Song Z, et al. Innate immune sensing of Z-nucleic acids by ZBP1-RIPK1 axis drives neuroinflammation in Alzheimer’s disease. Immunity. 2025;58(10):2574–2592.
- Glass CK, et al. Mechanisms underlying inflammation in neurodegeneration. Cell. 2010;140(6):918–934.
- Colonna M, Butovsky O. Microglia function in the central nervous system during health and neurodegeneration. Annu Rev Immunol. 2017;35:441–468.
- McGeer PL, McGeer EG. Glial reactions in Parkinson’s disease. Mov Disord. 2008;23(4):474–483.
- Philips T, Robberecht W. Neuroinflammation in amyotrophic lateral sclerosis: role of glial activation in motor neuron disease. Lancet Neurol. 2011;10(3):253–263.
- Jonsson T, et al. Variant of TREM2 associated with the risk of Alzheimer’s disease. N Engl J Med. 2013;368(2):107–116.
- Guerreiro R, et al. TREM2 variants in Alzheimer’s disease. N Engl J Med. 2013;368(2):117–127.
- Keren-Shaul H, et al. A unique microglia type associated with restricting development of Alzheimer’s disease. Cell. 2017;169(7):1276–1290.
- Mrdjen D, et al. High-dimensional single-cell mapping of central nervous system immune cells reveals distinct myeloid subsets in health, aging, and disease. Immunity. 2018;48(2):380–395.
- Zhou Y, et al. Human and mouse single-nucleus transcriptomics reveal TREM2-dependent and TREM2-independent cellular responses in Alzheimer’s disease. Nat Med. 2020;26(1):131–142.
- Kober DL, et al. Neurodegenerative disease mutations in TREM2 reveal a functional surface and distinct loss-of-function mechanisms. Elife. 2016;5:e20391.
- Song W, et al. Alzheimer’s disease-associated TREM2 variants exhibit either decreased or increased ligand-dependent activation. Alzheimers Dement. 2017;13(4):381–387.
- Wang S, et al. TREM2 drives microglia response to amyloid-β via SYK-dependent and -independent pathways. Cell. 2022;185(22):4153–4169.
- Ennerfelt H, et al. SYK coordinates neuroprotective microglial responses in neurodegenerative disease. Cell. 2022;185(22):4135–4152.
- Lee CYD, et al. Elevated TREM2 gene dosage reprograms microglia responsivity and ameliorates pathological phenotypes in Alzheimer’s disease models. Neuron. 2018;97(5):1032–1048.
- Wang Y, et al. TREM2 lipid sensing sustains the microglial response in an Alzheimer’s disease model. Cell. 2015;160(6):1061–1071.
- Wang Y, et al. TREM2-mediated early microglial response limits diffusion and toxicity of amyloid plaques. J Exp Med. 2016;213(5):667–675.
- Jay TR, et al. Disease progression-dependent effects of TREM2 deficiency in a mouse model of Alzheimer’s Disease. J Neurosci. 2017;37(3):637–647.
- Ulrich JD, et al. Altered microglial response to Aβ plaques in APPPS1-21 mice heterozygous for TREM2. Mol Neurodegener. 2014;9:20.
- Krasemann S, et al. The TREM2-APOE pathway drives the transcriptional phenotype of dysfunctional microglia in neurodegenerative diseases. Immunity. 2017;47(3):566–581.
- Xie M, et al. TREM2 interacts with TDP-43 and mediates microglial neuroprotection against TDP-43-related neurodegeneration. Nat Neurosci. 2022;25(1):26–38.
- Leyns CEG, et al. TREM2 deficiency attenuates neuroinflammation and protects against neurodegeneration in a mouse model of tauopathy. Proc Natl Acad Sci U S A. 2017;114(43):11524–11529.
- Sayed FA, et al. Differential effects of partial and complete loss of TREM2 on microglial injury response and tauopathy. Proc Natl Acad Sci U S A. 2018;115(40):10172–10177.
- Deczkowska A, et al. Disease-associated microglia: a universal immune sensor of neurodegeneration. Cell. 2018;173(5):1073–1081.
- Afify R, et al. Myeloid antigen-presenting cells in neurodegenerative diseases: a focus on classical and non-classical MHC molecules. Front Neurosci. 2024;18:1488382.
- Chen X, et al. Microglia-mediated T cell infiltration drives neurodegeneration in tauopathy. Nature. 2023;615(7953):668–677.
- Mason HD, et al. Granzyme K+ CD8 T cells slow tauopathy progression by targeting microglia. Nat Immunol. 2025;26(7):1152–1167.
- Harms AS, et al. MHCII is required for α-synuclein-induced activation of microglia, CD4 T cell proliferation, and dopaminergic neurodegeneration. J Neurosci. 2013;33(23):9592–9600.
- Rogers J, et al. Expression of immune system-associated antigens by cells of the human central nervous system: relationship to the pathology of Alzheimer’s disease. Neurobiol Aging. 1988;9(4):339–349.
- Itagaki S, et al. Presence of T-cytotoxic suppressor and leucocyte common antigen positive cells in Alzheimer’s disease brain tissue. Neurosci Lett. 1988;91(3):259–264.
- Joshi C, et al. CSF-derived CD4+ T-cell diversity is reduced in patients with Alzheimer clinical syndrome. Neurol Neuroimmunol Neuroinflamm. 2022;9(1):e1106.
- Ethell DW, et al. Abeta-specific T-cells reverse cognitive decline and synaptic loss in Alzheimer’s mice. Neurobiol Dis. 2006;23(2):351–361.
- Cao C, et al. Abeta-specific Th2 cells provide cognitive and pathological benefits to Alzheimer’s mice without infiltrating the CNS. Neurobiol Dis. 2009;34(1):63–70.
- Karikari AA, et al. Neurodegeneration by α-synuclein-specific T cells in AAV-A53T-α-synuclein Parkinson’s disease mice. Brain Behav Immun. 2022;101:194–210.
- Gate D, et al. CD4+ T cells contribute to neurodegeneration in Lewy body dementia. Science. 2021;374(6569):868–874.
- Sheean RK, et al. Association of regulatory T-cell expansion with progression of amyotrophic lateral sclerosis: a study of humans and a transgenic mouse model. JAMA Neurol. 2018;75(6):681–689.
- Brochard V, et al. Infiltration of CD4+ lymphocytes into the brain contributes to neurodegeneration in a mouse model of Parkinson disease. J Clin Invest. 2009;119(1):182–192.
- Iba M, et al. Neuroinflammation is associated with infiltration of T cells in Lewy body disease and α-synuclein transgenic models. J Neuroinflammation. 2020;17(1):214.
- Beers DR, et al. CD4+ T cells support glial neuroprotection, slow disease progression, and modify glial morphology in an animal model of inherited ALS. Proc Natl Acad Sci U S A. 2008;105(40):15558–15563.
- Chiu IM, et al. T lymphocytes potentiate endogenous neuroprotective inflammation in a mouse model of ALS. Proc Natl Acad Sci U S A. 2008;105(46):17913–17918.
- Williams GP, et al. CD4 T cells mediate brain inflammation and neurodegeneration in a mouse model of Parkinson’s disease. Brain. 2021;144(7):2047–2059.
- Sakaguchi S, et al. Regulatory T cells and immune tolerance. Cell. 2008;133(5):775–787.
- Ciccocioppo F, et al. The characterization of regulatory T-cell profiles in alzheimer’s disease and multiple sclerosis. Sci Rep. 2019;9(1):8788.
- Yazdani S, et al. T cell responses at diagnosis of amyotrophic lateral sclerosis predict disease progression. Nat Commun. 2022;13(1):6733.
- Saunders JA, et al. CD4+ regulatory and effector/memory T cell subsets profile motor dysfunction in Parkinson’s disease. J Neuroimmune Pharmacol. 2012;7(4):927–938.
- Merlini M, et al. Extravascular CD3+ T cells in brains of Alzheimer disease patients correlate with Tau but not with amyloid pathology: an immunohistochemical study. Neurodegener Dis. 2018;18(1):49–56.
- Unger MS, et al. CD8+ T-cells infiltrate Alzheimer’s disease brains and regulate neuronal- and synapse-related gene expression in APP-PS1 transgenic mice. Brain Behav Immun. 2020;89:67–86.
- Galiano-Landeira J, et al. CD8 T cell nigral infiltration precedes synucleinopathy in early stages of Parkinson’s disease. Brain. 2020;143(12):3717–3733.
- Laurent C, et al. Hippocampal T cell infiltration promotes neuroinflammation and cognitive decline in a mouse model of tauopathy. Brain. 2017;140(1):184–200.
- Kumar BV, et al. Human tissue-resident memory T cells are defined by core transcriptional and functional signatures in lymphoid and mucosal sites. Cell Rep. 2017;20(12):2921–2934.
- Altendorfer B, et al. Transcriptomic profiling identifies CD8+ T cells in the brain of aged and alzheimer’s disease transgenic mice as tissue-resident memory T cells. J Immunol. 2022;209(7):1272–1285.
- Su W, et al. CXCR6 orchestrates brain CD8+ T cell residency and limits mouse Alzheimer’s disease pathology. Nat Immunol. 2023;24(10):1735–1747.
- Piehl N, et al. Cerebrospinal fluid immune dysregulation during healthy brain aging and cognitive impairment. Cell. 2022;185(26):5028–5039.
- Hobson R, et al. Clonal CD8+ T cells populate the leptomeninges and coordinate with immune cells in human degenerative brain diseases. Nat Immunol. 2026;27(2):323–335.
- Muroyama Y, Wherry EJ. Memory T-cell heterogeneity and terminology. Cold Spring Harb Perspect Biol. 2021;13(10):a037929.
- van Olst L, et al. Adaptive immune changes associate with clinical progression of Alzheimer’s disease. Mol Neurodegener. 2024;19(1):38.
- Gate D, et al. Clonally expanded CD8 T cells patrol the cerebrospinal fluid in Alzheimer’s disease. Nature. 2020;577(7790):399–404.
- Capelle CM, et al. Early-to-mid stage idiopathic Parkinson’s disease shows enhanced cytotoxicity and differentiation in CD8 T-cells in females. Nat Commun. 2023;14(1):7461.
- Campisi L, et al. Clonally expanded CD8 T cells characterize amyotrophic lateral sclerosis-4. Nature. 2022;606(7916):945–952.
- Jonsson AH, et al. Granzyme K+ CD8 T cells form a core population in inflamed human tissue. Sci Transl Med. 2022;14(649):eabo0686.
- Donado CA, et al. Granzyme K activates the entire complement cascade. Nature. 2025;641(8061):211–221.
- Mogilenko DA, et al. Comprehensive profiling of an aging immune system reveals clonal GZMK+ CD8+ T cells as conserved hallmark of inflammaging. Immunity. 2021;54(1):99–115.
- Bouwman AC, et al. Intracellular and extracellular roles of granzyme K. Front Immunol. 2021;12:677707.
- Lan F, et al. GZMK-expressing CD8+ T cells promote recurrent airway inflammatory diseases. Nature. 2025;638(8050):490–498.
- Joeckel LT, et al. Granzyme K-deficient mice show no evidence of impaired antiviral immunity. Immunol Cell Biol. 2017;95(8):676–683.
- Terrabuio E, et al. CD103-CD8+ T cells promote neurotoxic inflammation in Alzheimer’s disease via granzyme K-PAR-1 signaling. Nat Commun. 2025;16(1):8372.
- Kim HJ, et al. Single-cell analysis reveals expanded CD8+GZMKhigh T cells in CSF and shared peripheral clones in sporadic amyotrophic lateral sclerosis. Brain Commun. 2024;6(6):fcae428.
- Franceschi C, et al. Inflammaging: a new immune-metabolic viewpoint for age-related diseases. Nat Rev Endocrinol. 2018;14(10):576–590.
- Pinti M, et al. Circulating mitochondrial DNA increases with age and is a familiar trait: Implications for “inflamm-aging”. Eur J Immunol. 2014;44(5):1552–1562.
- Gulen MF, et al. cGAS-STING drives ageing-related inflammation and neurodegeneration. Nature. 2023;620(7973):374–380.
- da Fonseca AC, et al. The impact of microglial activation on blood-brain barrier in brain diseases. Front Cell Neurosci. 2014;8:362. View this article via: PubMed Google Scholar
- Nyul-Toth A, et al. Demonstration of age-related blood-brain barrier disruption and cerebromicrovascular rarefaction in mice by longitudinal intravital two-photon microscopy and optical coherence tomography. Am J Physiol Heart Circ Physiol. 2021;320(4):1370–1392.
- Sweeney MD, et al. Blood-brain barrier: from physiology to disease and back. Physiol Rev. 2019;99(1):21–78.
- Groh J, et al. Accumulation of cytotoxic T cells in the aged CNS leads to axon degeneration and contributes to cognitive and motor decline. Nat Aging. 2021;1(4):357–367.
- Sun ED, et al. Spatial transcriptomic clocks reveal cell proximity effects in brain ageing. Nature. 2025;638(8049):160–171.
- Li Y, et al. Head injury as a risk factor for dementia and Alzheimer’s disease: a systematic review and meta-analysis of 32 observational studies. PLoS One. 2017;12(1):e0169650.
- Fann JR, et al. Long-term risk of dementia among people with traumatic brain injury in Denmark: a population-based observational cohort study. Lancet Psychiatry. 2018;5(5):424–431.
- Nordstrom A, Nordstrom P. Traumatic brain injury and the risk of dementia diagnosis: A nationwide cohort study. PLoS Med. 2018;15(1):e1002496.
- Sariaslan A, et al. Long-term outcomes associated with traumatic brain injury in childhood and adolescence: a nationwide Swedish cohort study of a wide range of medical and social outcomes. PLoS Med. 2016;13(8):e1002103.
- Walko TD, et al. Cerebrospinal fluid mitochondrial DNA: a novel DAMP in pediatric traumatic brain injury. Shock. 2014;41(6):499–503.
- Corps KN, et al. Inflammation and neuroprotection in traumatic brain injury. JAMA Neurol. 2015;72(3):355–362.
- Russo MV, McGavern DB. Inflammatory neuroprotection following traumatic brain injury. Science. 2016;353(6301):783–785.
- Roth TL, et al. Transcranial amelioration of inflammation and cell death after brain injury. Nature. 2014;505(7482):223–228.
- Nayak D, et al. Microglia development and function. Annu Rev Immunol. 2014;32:367–402.
- Mason HD, et al. Glia limitans superficialis oxidation and breakdown promote cortical cell death after repetitive head injury. JCI Insight. 2021;6(19):e149229.
- Mastorakos P, et al. Temporally distinct myeloid cell responses mediate damage and repair after cerebrovascular injury. Nat Neurosci. 2021;24(2):245–258.
- McKee AC, et al. The first NINDS/NIBIB consensus meeting to define neuropathological criteria for the diagnosis of chronic traumatic encephalopathy. Acta Neuropathol. 2016;131(1):75–86.
- McKee AC, et al. The spectrum of disease in chronic traumatic encephalopathy. Brain. 2013;136(pt 1):43–64.
- McKee AC, et al. The neuropathology of chronic traumatic encephalopathy. Brain Pathol. 2015;25(3):350–364.
- Mez J, et al. Clinicopathological evaluation of chronic traumatic encephalopathy in players of american football. JAMA. 2017;318(4):360–370.
- Ojo JO, et al. Chronic repetitive mild traumatic brain injury results in reduced cerebral blood flow, axonal injury, gliosis, and increased T-Tau and Tau oligomers. J Neuropathol Exp Neurol. 2016;75(7):636–655.
- Washington PM, et al. Experimental traumatic brain injury induces rapid aggregation and oligomerization of amyloid-beta in an Alzheimer’s disease mouse model. J Neurotrauma. 2014;31(1):125–134.
- Shishido H, et al. Traumatic brain injury accelerates amyloid-β deposition and impairs spatial learning in the triple-transgenic mouse model of Alzheimer’s disease. Neurosci Lett. 2016;629:62–67.
- Wu D, et al. Traumatic brain injury accelerates the onset of cognitive dysfunction and aggravates Alzheimer’s-like pathology in the hippocampus by altering the phenotype of microglia in the APP/PS1 mouse model. Front Neurol. 2021;12:666430.
- Petraglia AL, et al. The pathophysiology underlying repetitive mild traumatic brain injury in a novel mouse model of chronic traumatic encephalopathy. Surg Neurol Int. 2014;5:184.
- Schultz JL, et al. Human immunodeficiency virus infection in Huntington’s disease is associated with an earlier age of symptom onset. J Huntingtons Dis. 2018;7(2):163–166.
- Gonzalez H, et al. Identification of novel candidate protein biomarkers for the post-polio syndrome - implications for diagnosis, neurodegeneration and neuroinflammation. J Proteomics. 2009;71(6):670–681.
- Li C, et al. COVID-19 and risk of neurodegenerative disorders: A Mendelian randomization study. Transl Psychiatry. 2022;12(1):283.
- Wiley CA. Viral infection and dementia: A brief synthesis. Free Neuropathol. 2021;2:15. View this article via: PubMed Google Scholar
- Levine KS, et al. Virus exposure and neurodegenerative disease risk across national biobanks. Neuron. 2023;111(7):1086–1093.
- Swanson PA, et al. Viral diseases of the central nervous system. Curr Opin Virol. 2015;11:44–54.
- Arsenio-Nunes ML, et al. Vascular and neuroglial changes in experimental herpes simplex encephalitis: ultrastructural study. Acta Neuropathol. 1975;33(3):245–256.
- Eugenin EA, et al. CCL2/monocyte chemoattractant protein-1 mediates enhanced transmigration of human immunodeficiency virus (HIV)-infected leukocytes across the blood-brain barrier: a potential mechanism of HIV-CNS invasion and NeuroAIDS. J Neurosci. 2006;26(4):1098–1106.
- Drokhlyansky E, et al. The brain parenchyma has a type I interferon response that can limit virus spread. Proc Natl Acad Sci U S A. 2017;114(1):E95–E104.
- Taquet M, et al. The recombinant shingles vaccine is associated with lower risk of dementia. Nat Med. 2024;30(10):2777–2781.
- Eyting M, et al. A natural experiment on the effect of herpes zoster vaccination on dementia. Nature. 2025;641(8062):438–446.
- Xie M, et al. The effect of shingles vaccination at different stages of the dementia disease course. Cell. 2025;188(25):7049–7064.
- Smolders J, et al. Tissue-resident memory T cells populate the human brain. Nat Commun. 2018;9(1):4593.
- Shrestha B, et al. Gamma interferon plays a crucial early antiviral role in protection against West Nile virus infection. J Virol. 2006;80(11):5338–5348.
- McGavern DB, Kang SS. Illuminating viral infections in the nervous system. Nat Rev Immunol. 2011;11(5):318–329.
- Urban SL, et al. Peripherally induced brain tissue-resident memory CD8+ T cells mediate protection against CNS infection. Nat Immunol. 2020;21(8):938–949.
- Mix MR, et al. Repetitive antigen stimulation in the periphery dictates the composition and recall responses of brain-resident memory CD8+ T cells. Cell Rep. 2025;44(2):115247.
- Garber C, et al. T cells promote microglia-mediated synaptic elimination and cognitive dysfunction during recovery from neuropathogenic flaviviruses. Nat Neurosci. 2019;22(8):1276–1288.
- Wozniak MA, et al. Herpes simplex virus type 1 DNA is located within Alzheimer’s disease amyloid plaques. J Pathol. 2009;217(1):131–138.
- Eimer WA, et al. Alzheimer’s disease-associated β-amyloid is rapidly seeded by herpesviridae to protect against brain infection. Neuron. 2018;99(1):56–63.
- Bocharova O, et al. Alzheimer’s disease-associated β-amyloid does not protect against herpes simplex virus 1 infection in the mouse brain. J Biol Chem. 2021;297(1):100845.
-
Version history
- Version 1 (April 15, 2026): Electronic publication
Article tools
-
View PDF

- Download citation information
- Send a comment
- Terms of use
- Standard abbreviations
- Need help? Email the journal
Review Series
Neurodegeneration
-
Women’s midlife: the front line of Alzheimer prevention
Lisa Mosconi -
Genetic analysis of neurodegenerative diseases
Maurizio Grassano et al. -
Decoding neurodegeneration one cell at a time
Olivia Gautier et al. -
Lysosomal homeostasis at the crossroads of neurodegeneration
Stefano De Tito et al. -
Splicing the narrative: alternative TARDBP splicing and its relation to neurodegeneration in ALS and FTD
Morgan R. Miller et al. -
Immune signaling and function in neurodegeneration
Yvonne L. Latour et al.
Metrics



Copyright © 2026 American Society for Clinical Investigation
ISSN: 0021-9738 (print), 1558-8238 (online)



