Advertisement
Commentary
Open Access |  10.1172/JCI202410
10.1172/JCI202410
Aminopeptidase N: the glucocorticoid gateway linking chronic stress to ferroptosis resistance in liver cancer
Maowu Luo1 and Weibo Luo1,2
1Department of Pathology and
2Department of Pharmacology, UT Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Weibo Luo, Department of Pathology, UT Southwestern Medical Center, 5323 Harry Hines Blvd., NB6.460, Dallas, Texas, 75390, USA. Phone: 214.645.4770; Email: Weibo.Luo@UTSouthwestern.edu.
Find articles by Luo, M. in: PubMed | Google Scholar
1Department of Pathology and
2Department of Pharmacology, UT Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Weibo Luo, Department of Pathology, UT Southwestern Medical Center, 5323 Harry Hines Blvd., NB6.460, Dallas, Texas, 75390, USA. Phone: 214.645.4770; Email: Weibo.Luo@UTSouthwestern.edu.
Find articles by
Luo, W.
in:
PubMed
|
Google Scholar
|

Published February 16, 2026 - More info
J Clin Invest. 2026;136(4):e202410. https://doi.org/10.1172/JCI202410.
© 2026 Luo et al. This work is licensed under the Creative Commons Attribution 4.0 International License. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
Related article:
Abstract
Emerging evidence demonstrates that chronic stress alters immunological, neurochemical, and endocrinological functions, thereby promoting tumor progression. However, the underlying metabolic mechanism of chronic stress in tumor progression is still elusive. Using multiomics analysis, we found that aminopeptidase N (ANPEP) was upregulated in tumors with chronic restraint, associating with the reprogramming of amino acid metabolism. Functional assays revealed that ANPEP promoted liver cancer growth and metastasis. Knockdown of ANPEP blocked chronic stress–induced liver cancer progression. Chronic stress–induced glucocorticoids promoted nuclear receptor subfamily 3 group C member 1 nuclear translocation to activate ANPEP transcription by directly binding to its promoter. Furthermore, ANPEP promotes glutathione synthesis, subsequently inhibiting ROS-induced ferroptosis. Mechanistically, ANPEP interacted with solute carrier family 3 member 2 (SLC3A2) to block membrane associated ring-CH-type finger 8–mediated lysosome-dependent degradation of SLC3A2, promoting intracellular l-cystine transport, thereby increasing glutathione synthesis. The combination of ANPEP silencing and sorafenib treatment showed a synergistic effect in inhibiting liver cancer progression. Finally, clinical data and mouse models demonstrated that chronic stress drove liver tumor progression via ANPEP-regulated SLC3A2. These findings reveal unanticipated communication between chronic stress and metabolic reprogramming during liver cancer progression, providing potential therapeutic implications for liver cancer.
Authors
Yongkang Wu, Yankun Zhang, Xiaojia Shi, Mengting Wu, Min Sun, Ying Feng, Wenmeng Ma, Xiule Jiang, Dingqi Fei, Mingjian Zhao, Zhuanchang Wu, Chunyang Li, Xiaohong Liang, Lifen Gao, Chunhong Ma, Xuetian Yue
-
Abstract
Chronic stress triggers a range of physiological responses that could dysregulate the immune system and metabolic processes, thereby increasing susceptibility to various diseases. In this issue of the JCI, Wu et al. identified a metabolic bridge between chronic stress and liver cancer progression. Chronic stress–induced glucocorticoids promoted aminopeptidase N (ANPEP) expression and subsequent reprogramming of amino acid metabolism, leading to increased liver cancer growth and metastasis. ANPEP facilitated stabilization of the cystine-glutamate transporter system Xc– and increased l-cystine influx, thereby enhancing cellular antioxidant capacity to prevent ferroptosis. Silencing ANPEP in combination with sorafenib treatment showed a synergistic inhibitory effect on liver cancer progression. These findings uncover ANPEP as a valuable target for therapeutic interventions to treat patients with liver cancer experiencing chronic stress.
-
Chronic stress and liver cancer
Liver cancer develops after chronic liver injuries, including cirrhosis, hepatitis B or C virus infection, alcohol use, and metabolic dysfunction–associated steatohepatitis (1). It is a malignant disease with a steadily increasing incidence and mortality in the United States (2). Recent clinical studies reveal that chronic stress is one of the factors associated with poor survival outcomes in patients with liver cancer (3). Chronic stress, a common psychological condition in modern society, arises from prolonged exposure to stressors in daily life and has detrimental effects on physical and mental health (4). In contrast with acute stress, chronic stress sustains a prolonged activation of the hypothalamic-pituitary-adrenal axis and the sympathetic nervous system, resulting in a continuous release of glucocorticoids (GCs) and catecholamines (Figure 1A) (5). Previous studies have shown that these stress hormones promote cancer development and metastasis by regulating angiogenesis, metabolism, DNA damage, inflammation, and immunosuppression in various preclinical cancer models (6–10). However, the contribution of chronic stress to liver cancer and the mechanisms involved remain poorly defined.
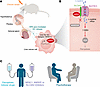 Figure 1
Figure 1Chronic stress–induced ANPEP drives ferroptosis resistance in liver cancer. (A) Wu et al. (11) modeled chronic stress–mediated activation of the hypothalamic-pituitary-adrenal axis using chronic restraint stress. Stress-induced GCs mediated nuclear translocation of NR3C1, activating ANPEP transcription in liver cancer cells. (B) Wu et al. showed that ANPEP interacted with SLC3A2, a transmembrane subunit of the l-cystine/glutamate antiporter system Xc– (which also contains the subunit SLC7A11 shown in the figure). This interaction prevented SLC3A2 from undergoing MARCH8-mediated lysosome-dependent degradation, thereby increasing synthesis of the intracellular antioxidant GSH and protecting liver cancer cells against ferroptosis. (C) Potential therapeutic strategies for liver cancer may include targeting the NR3C1/ANPEP/SLC3A2 axis or implementing psychotherapy to reduce stress, sensitizing tumors to ferroptosis-inducing anticancer therapies.
In this issue of the JCI, Wu et al. (11) identified a detailed mechanism that elucidated how stress hormones promoted liver cancer progression and metastasis (Figure 1, A and B). The authors used a chronic restraint stress murine model that dramatically elevates serum corticosterone (the murine GC), demonstrating a remarkable effect of chronic stress on liver cancer progression in this animal model. Utilizing multiomics, including metabolomics and RNA sequencing, they identified a consequential rewiring of amino acid metabolic pathways, with Anpep emerging as the most significantly upregulated gene. Anpep encodes aminopeptidase N, an enzyme involved in amino acid metabolism. Next, in two human liver cancer cell lines, Wu et al. uncovered the key transcriptional mechanism underlying ANPEP upregulation: Upon GC stimulation, nuclear receptor subfamily 3 group C member 1 (NR3C1) directly bound to the ANPEP promoter and enhanced its transcription (Figure 1A). The GC/NR3C1/ANPEP axis acts as a molecular bridge, translating systemic chronic stress into a specific, actionable metabolic command within the liver cancer cell. This finding transforms ANPEP from a mere prognostic marker into a GC-responsive metabolic gateway in liver cancer (12, 13). The observation that ANPEP was also upregulated by chronic stress in colon cancer suggests that this mechanism may represent a generalizable pathway linking chronic stress to tumor-specific metabolic reprogramming across multiple cancer types (11).
-
ANPEP upregulation promotes ferroptosis resistance in liver cancer
Cancer cells inherently face high levels of oxidative stress because of their rapid proliferation, high metabolic activity, and harsh tumor microenvironment (14). Oxidative stress causes lipid peroxidation and triggers ferroptosis, an iron-dependent nonapoptotic form of cell death (15). While the liver serves as a major iron reservoir (16), liver cancer cells adapt to redox imbalance by activating antioxidant defense systems, thereby limiting ferroptosis (17). Glutathione (GSH), an abundant cellular antioxidant, plays a pivotal role in this process (18). The authors convincingly showed that ANPEP, once transcriptionally activated by chronic stress, promoted GSH synthesis to reduce reactive oxygen species and lipid peroxidation in liver cancer cells, thereby preventing ferroptosis and conferring a critical survival advantage (11).
GSH is a tripeptide composed of three amino acids: l-cysteine, l-glutamate, and l-glycine. The availability of l-cysteine is the rate-limiting step in GSH synthesis. The stable influx of l-cystine via the cystine/glutamate antiporter system Xc– and its subsequent oxidation to l-cysteine are critical to sustain high GSH levels (18, 19). In the present work, Wu et al. showed that ANPEP interacted with and stabilized solute carrier family 3 member 2 (SLC3A2) protein (11), a transmembrane subunit of system Xc– (Figure 1B). This interaction prevented the E3 ubiquitin ligase membrane associated ring-CH-type finger 8 (MARCH8) from binding and ubiquitinating SLC3A2, thereby inhibiting its lysosomal trafficking and degradation (Figure 1B). Through its interaction with SLC3A2, ANPEP is also associated with SLC7A11, another subunit of system Xc– responsible for l-cystine influx (19). Consequently, ANPEP promoted chronic stress–induced l-cystine uptake, leading to increased intracellular l-cysteine and elevated GSH levels in liver cancer cells (Figure 1B). This signaling cascade protected liver cancer cells against oxidative stress and ferroptosis (Figure 1B).
Experimental therapeutic studies showed that genetic inhibition of ANPEP or SLC3A2 prevented chronic stress–induced liver cancer xenograft growth and metastasis in mice (11). Notably, ANPEP overexpression reduced liver cancer sensitivity to sorafenib, an FDA-approved liver cancer drug known to induce ferroptosis (20), whereas ANPEP silencing enhanced sorafenib’s antitumor efficacy in mice by increasing intratumoral lipid peroxidation and ferroptosis (11). These findings provide a strong rationale for targeting cellular metabolism in combination with standard therapy to treat liver cancer (Figure 1C).
-
Impact and future directions
The findings by Wu et al. provide innovative insights into psychological regulation of metabolism–cell fate crosstalk that drives cancer development. The chronic stress signature the authors developed to predict survival outcomes in patients with liver cancer represents a clinically important prognostic tool. More importantly, this study highlights the potential of specific ANPEP or SLC3A2 inhibitors as therapeutic interventions for liver cancer, particularly for patients with elevated stress hormone levels.
Although comprehensive in its identification of a pathway linking chronic stress to liver cancer progression, the study raises several outstanding questions to be addressed in the future. (a) People are exposed to various forms of psychological stress in modern society, and chronic restraint stress may model specific types of stress. It remains unclear whether ANPEP-mediated signaling similarly contributes to tumor progression and metastasis under other forms of psychological stress, such as those modeled by social isolation. (b) ANPEP is a transmembrane peptidase that plays diverse roles in peptide digestion, antigen presentation, and cell signaling (21). Future investigation should determine whether its enzymatic activity, which hydrolyzes amino acids and peptides, is responsible for the metabolic reprogramming observed in this study. (c) Amino acids serve as essential fuels for tumor progression (22). Considering that multiple amino acid pathways are altered by chronic stress and ANPEP (11), a comprehensive understanding of the ANPEP-dependent metabolic landscape in response to chronic stress may provide critical insights into how chronic stress reshapes tumor metabolism and identify additional therapeutic opportunities. (d) Psychotherapy has been shown to alleviate chronic stress (23). It would be interesting to study whether psychotherapy can improve outcomes in patients with liver cancer receiving standard therapies (Figure 1C).
In conclusion, in this JCI paper, Wu et al. elucidate a fundamental metabolic connection between chronic stress and liver cancer progression. By identifying the ANPEP/SLC3A2 axis as a key mediator of chronic stress–induced ferroptosis resistance and liver cancer progression, the study offers a major conceptual advance as well as a strong rationale for developing new therapeutic strategies for liver cancer.
-
Funding support
- Grants from Department of Defense (HT9425-23-1-0863, HT9425-24-1-0538, and HT9425-24-1-0702).
- CPRIT (grant RP220178).
- CPRIT Scholar in Cancer Research (WL).
- Alex’s Lemonade Stand Foundation (grant 1433529).
-
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Copyright: © 2026, Luo et al. This is an open access article published under the terms of the Creative Commons Attribution 4.0 International License.
Reference information: J Clin Invest. 2026;136(4):e202410. https://doi.org/10.1172/JCI202410.
See the related article at Chronic stress–induced ANPEP drives liver cancer progression by increasing glutathione synthesis and inhibiting ferroptosis.
-
References
- Villanueva A. Hepatocellular carcinoma. N Engl J Med. 2019;380(15):1450–1462.
- Yao Z, et al. Time-trends in liver cancer incidence and mortality rates in the U.S. from 1975 to 2017: a study based on the surveillance, epidemiology, and end results database. J Gastrointest Oncol. 2023;14(1):312–324.
- Wang X, et al. Chronic stress impacts the prognosis of hepatocellular carcinoma patients after curative treatment by establishing a novel comprehensive classification: a cohort study and systematic review. BMC Psychiatry. 2025;25(1):937.
- Eckerling A, et al. Stress and cancer: directions. Nat Rev Cancer. 2021;21(12):767–785.
- Russell G, Lightman S. The human stress response. Nat Rev Endocrinol. 2019;15(9):525–534.
- He XY, et al. Chronic stress increases metastasis via neutrophil-mediated changes to the microenvironment. Cancer Cell. 2024;42(3):474–486.
- Globig AM, et al. The β1-adrenergic receptor links sympathetic nerves to T cell exhaustion. Nature. 2023;622(7982):383–392.
- Thaker PH, et al. Chronic stress promotes tumor growth and angiogenesis in a mouse model of ovarian carcinoma. Nat Med. 2006;12(8):939–944.
- Li G, et al. Wip1 contributes to the adaptation of HepG2 human liver cancer cells to stress hormone-induced DNA damage. Oncol Lett. 2023;25(1):31.
- Wang T, et al. Chronic stress-induced cholesterol metabolism abnormalities promote ESCC tumorigenesis and predict neoadjuvant therapy response. Proc Natl Acad Sci U S A. 2025;122(5):e2415042122.
- Wu Y, et al. Chronic stress–induced ANPEP drives liver cancer progression by increasing glutathione synthesis and inhibiting ferroptosis. J Clin Invest. 2026;136(4):e195685.
- Barnieh FM, et al. Is tumour-expressed aminopeptidase N (APN/CD13) structurally and functionally unique? Biochim Biophys Acta Rev Cancer. 2021;1876(2):188641.
- Rocken C, et al. Canalicular immunostaining of aminopeptidase N (CD13) as a diagnostic marker for hepatocellular carcinoma. J Clin Pathol. 2005;58(10):1069–1075.
- Pillai R, et al. NRF2: KEAPing tumors protected. Cancer Discov. 2022;12(3):625–643.
- Capelletti MM, et al. Ferroptosis in liver diseases: an overview. Int J Mol Sci. 2020;21(14):4908.
- Anderson ER, Shah YM. Iron homeostasis in the liver. Compr Physiol. 2013;3(1):315–330.
- Raghunath A, et al. Dysregulation of Nrf2 in hepatocellular carcinoma: role in cancer progression and chemoresistance. Cancers (Basel). 2018;10(12):481.
- Bansal A, Simon MC. Glutathione metabolism in cancer progression and treatment resistance. J Cell Biol. 2018;217(7):2291–2298.
- Bridges RJ, et al. System Xc– cystine/glutamate antiporter: an update on molecular pharmacology and roles within the CNS. Br J Pharmacol. 2012;165(1):20–34.
- Sun J, et al. Quiescin sulfhydryl oxidase 1 promotes sorafenib-induced ferroptosis in hepatocellular carcinoma by driving EGFR endosomal trafficking and inhibiting NRF2 activation. Redox Biol. 2021;41:101942.
- Lendeckel U, et al. The role of the ectopeptidase APN/CD13 in cancer. Biomedicines. 2023;11(3):724.
- Chen J, et al. Amino acid metabolism in tumor biology and therapy. Cell Death Dis. 2024;15(1):42.
- Cruess DG, et al. Cognitive-behavioral stress management reduces serum cortisol by enhancing benefit finding among women being treated for early stage breast cancer. Psychosom Med. 2000;62(3):304–308.
-
Version history
- Version 1 (February 16, 2026): Electronic publication



Copyright © 2026 American Society for Clinical Investigation
ISSN: 0021-9738 (print), 1558-8238 (online)

