Advertisement
Research ArticleCardiologyMetabolism
Open Access |  10.1172/JCI202528
10.1172/JCI202528
Unrestrained fatty acid oxidation triggers heart failure in mice via cardiolipin loss and mitochondrial dysfunction
Chai-Wan Kim,1,2 Goncalo Vale,1,2 Xiaorong Fu,1 Jeffrey G. McDonald,1,3 Chongshan Dai,4 Chao Li,4,5 Zhao V. Wang,4,6 Gaurav Sharma,7,8 Chalermchai Khemtong,8 Craig R. Malloy,5 Stanislaw Deja,1 Shawn C. Burgess,1 Matthew A. Mitsche,1 and Jay D. Horton1,2,3
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Kim, C. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Vale, G. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Fu, X. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by
McDonald, J.
in:
PubMed
|
Google Scholar
|

1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Dai, C. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Li, C. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Wang, Z. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by
Sharma, G.
in:
PubMed
|
Google Scholar
|

1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Khemtong, C. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Malloy, C. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by
Deja, S.
in:
PubMed
|
Google Scholar
|

1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Burgess, S. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by Mitsche, M. in: PubMed | Google Scholar
1Center for Human Nutrition,
2Department of Internal Medicine,
3Department of Molecular Genetics,
4Division of Cardiology, and
5Touchstone Diabetes Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
6Department of Diabetes and Cancer Metabolism, Beckman Research Institute, City of Hope National Medical Center, Duarte, California, USA.
7Department of Cardiovascular and Thoracic Surgery and
8Advanced Imaging Research Center, University of Texas Southwestern Medical Center, Dallas, Texas, USA.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
Find articles by
Horton, J.
in:
PubMed
|
Google Scholar
|

Published May 1, 2026 - More info
J Clin Invest. 2026;136(9):e202528. https://doi.org/10.1172/JCI202528.
© 2026 Kim et al. This work is licensed under the Creative Commons Attribution 4.0 International License. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
Received: November 17, 2025; Accepted: March 10, 2026
Related article:
Abstract
Fatty acid oxidation (FAO) provides the healthy heart with 60%–90% of its ATP, with the remainder coming from metabolism of glucose. Metabolic flexibility is key to heart function, ensuring an uninterrupted source of fuel. In heart failure, a shift from FAO to glucose-dependent metabolism occurs as disease progresses, supporting the widely held notion that fat is the optimal substrate in the heart. In this issue of the JCI, Kim et al. challenge this assumption. In studies of acetyl-CoA carboxylase–deficient (ACC-deficient) mice, they found that unregulated use of fat as a substrate led to cardiac damage. ACC-deficient mice developed cardiolipin deficiency as a result of excessive FAO depleting stores of linoleic acid, which is used as a substrate for cardiolipin maturation. The resulting mitochondrial dysfunction was associated with dilated cardiomyopathy and heart failure in these mice. The findings highlight potential for development of therapeutic strategies that balance energy sources and replenish cardiolipin levels.
Authors
Steven M. Claypool, Carla M. Koehler
-
Abstract
Cardiomyocytes primarily rely on fatty acid oxidation (FAO), which provides more than 70% of their energy. However, excessive FAO can disrupt cardiac metabolism by increasing oxygen demand and suppressing glucose utilization through the Randle cycle. Although inhibition of FAO has been investigated in heart failure, its overall therapeutic impact remains uncertain. To determine the consequences of enhanced FAO, we generated cardiomyocyte-specific ACC1 and ACC2 double-knockout (ACC dHKO) mice, which exhibit constitutively elevated FAO. ACC dHKO mice developed dilated cardiomyopathy and heart failure. Lipidomic analysis revealed marked depletion of cardiolipin caused by reduced linoleic acid, a direct consequence of excessive FAO. This cardiolipin deficiency impaired mitochondrial electron transport chain (ETC) activity, leading to mitochondrial dysfunction. Pharmacologic inhibition of FAO with etomoxir or oxfenicine restored cardiolipin levels, normalized ETC activity, and prevented cardiac dysfunction in ACC dHKO mice. These findings demonstrate that unrestrained FAO disrupts both lipid and energy homeostasis, culminating in heart failure in this model. Collectively, these results indicate that although FAO is essential for cardiac energy production, therapeutic strategies aimed at stimulating cardiac FAO may be detrimental rather than beneficial in heart failure.
-
Introduction
Cardiomyopathy is a prevalent and debilitating form of heart failure that encompasses a diverse group of myocardial disorders characterized by structural and functional abnormalities, often leading to compromised cardiac function and, in many cases, heart failure (1–3). Currently, heart failure affects approximately 6.2 million people in the United States (4), with cardiomyopathy being a leading underlying cause (5). Moreover, the incidence of cardiomyopathy is rising, driven by various risk factors, including obesity, hypertension, and diabetes (6–8).
Recent studies have emphasized the pivotal role that alterations in cardiac energy metabolism play in the pathogenesis of cardiomyopathy (reviewed in refs. 9, 10). Remarkably, the human heart consumes approximately 6 kg of ATP daily. More than 95% of this ATP is supplied through the oxidative phosphorylation of fatty acids (FAs) and glucose (2, 11). In a healthy heart, 60%–90% of ATP is generated through FA oxidation (FAO), while the contribution of glucose oxidation is 10%–40% (10). However, in heart failure, the proportion of FAO decreases relative to glucose utilization as the disease progresses (1, 2). Since FAO requires 10%–15% more oxygen than glucose oxidation, the decrease in FAO may be a compensatory mechanism of the failing heart to conserve oxygen (12, 13). Although there is substantial debate as to whether changes in the ratio of FA and glucose oxidation are causally linked to the development of heart failure (10, 13, 14), several clinical studies have suggested that partial inhibition of FAO has therapeutic potential for heart failure (15, 16).
In recent decades, insights into the intricate interplay between FA and glucose oxidation and its profound impact on the development of cardiomyopathy have been revealed (10). A key regulatory enzyme that determines substrate utilization within the heart is acetyl-CoA carboxylase (ACC). ACC is responsible for the conversion of acetyl-CoA to malonyl-CoA, a molecule with dual functions (17). Malonyl-CoA serves as a building block for FA synthesis (18) and as a regulator of FAO by inhibiting carnitine palmitoyltransferase 1 (CPT1), the transporter of long-chain FAs into mitochondria (19). By modulating FAO, ACC influences the balance between FA and glucose oxidation. All cells express 2 forms of ACC. ACC1 is in the cytosol and produces malonyl-CoA for FA synthesis. ACC2 is associated with the mitochondrial membrane and produces malonyl-CoA primarily to regulate CPT1 (20). Cardiomyocytes have very low rates of de novo lipogenesis, and the expression of ACC1 is very low compared with lipogenic organs such as the liver (20, 21). In contrast, the expression of ACC2 in cardiomyocytes is very high compared with most other tissues (21).
Mouse models with altered ACC expression in various tissues have provided compelling evidence that dysregulation of this enzyme is associated with changes in FA and glucose oxidation (18, 22, 23). A complete understanding of the intricate role of ACCs in cardiac energy metabolism may open new avenues for developing tailored approaches to prevent or manage cardiomyopathy and improve patient outcomes.
In this study, deleting ACC1 and ACC2 in cardiomyocytes led to unrestrained FAO and a significant reduction of cardiolipin, causing mitochondrial dysfunction and ultimately heart failure. These deleterious effects were prevented in mice lacking ACC1 and ACC2 by administering drugs that suppress FAO. These findings highlight the potential risk of elevated FAO and urge caution in adapting strategies aimed at promoting FAO in heart failure.
-
Results
Cardiomyocyte-specific ACC1 (ACC1 HKO), ACC2 (ACC2 HKO), and double-knockout (ACC dHKO) mice were generated by crossing Acc1- and Acc2-floxed alleles with Myh6-Cre transgenic mice (18). Deletions of Acc1 and Acc2 were verified by qPCR (Figure 1, A and B). Acc1 mRNA levels were reduced by approximately 60% in ACC1 HKO and ACC dHKO hearts, while Acc2 mRNA levels were reduced by approximately 95% in ACC2 HKO and ACC dHKO hearts. As indicated by qPCR Ct values, basal Acc1 expression in heart was substantially lower than Acc2, consistent with a previous report (24). The residual Acc1 and Acc2 mRNA detected in the respective cardiomyocyte-specific knockouts was likely due to noncardiomyocyte expression within whole-heart RNA preparations. To confirm functional loss of ACC activity, we measured malonyl-CoA, the enzymatic product of ACC (Figure 1C). Total malonyl-CoA concentrations were approximately 89% lower in ACC dHKO hearts than in WT controls, whereas acetyl-CoA (the substrate of ACC) was unchanged across genotypes.
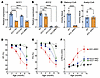 Figure 1
Figure 1Genetic deletion of ACC1 and ACC2 in mouse cardiomyocytes. (A and B) Total RNA was isolated from hearts of 10-week-old WT, ACC1 HKO, ACC2 HKO, and ACC dHKO male mice, and RT-qPCR was performed to measure Acc1 and Acc2 mRNA expression (n = 6 WT, 5 ACC1 HKO, 2 ACC2 HKO, and 5 ACC dHKO for A; n = 6 WT, 5 ACC1 HKO, 2 ACC2 HKO, and 5 ACC dHKO for B). ***P < 0.001, ****P < 0.0001 by 1-way ANOVA. (C) Hearts from 20-week-old WT and ACC dHKO female mice were harvested and freeze-clamped to measure malonyl-CoA and acetyl-CoA levels, as described in Methods (n = 6 WT and 7 ACC dHKO). ****P < 0.0001 by unpaired 2-tailed Student’s t test. (D–F) ECHO assessment of cardiac function in male ACC1 HKO, ACC2 HKO, and ACC dHKO mice at 2, 4, 6, and 9 months of age (n = 10 WT, 5 ACC1 HKO, 7 ACC2 HKO, and 5 ACC dHKO at 2, 4, and 6 months; n = 10 WT, 5 ACC1 HKO, 7 ACC2 HKO, and 4 ACC dHKO at 9 months). Data are presented as the mean ± SEM.
Cardiac function was assessed by echocardiography (ECHO). ACC1 HKO mice exhibited normal cardiac function up to approximately 6 months, but by 9 months exhibited a 20% reduction in ejection fraction (EF) and a 34% reduction in fractional shortening (FS) relative to WT (Figure 1, D and E). ACC2 HKO mice developed mild dysfunction by 6 months (~10% decrease in EF and FS) that progressed to 29% and 42% reductions in EF and FS, respectively, by 9 months (Figure 1, D and E). In contrast, ACC dHKO mice exhibited systolic dysfunction by 2 months, with increased left ventricular internal diameter (LVID) during systole and reduced EF and FS (Figure 1F). Cardiac function then progressively declined, with EF and FS falling to 40% and 53% of WT, respectively, by 9 months. Together, these data demonstrate that combined loss of ACC1 and ACC2 in cardiomyocytes leads to early-onset and progressive cardiomyopathy in this model, whereas deletion of either isoform alone results in delayed or milder impairment.
Given the potential functional overlap between ACC1 and ACC2 in cardiomyocytes, we further characterized ACC dHKO mice to define mechanisms leading to heart failure in this model. H&E staining revealed marked left ventricular enlargement in 11-month-old ACC dHKO hearts compared with WT controls (Figure 2A). Consistent with this, ECHO demonstrated increased LVID during both systole and diastole, accompanied by reduced left ventricular posterior wall thickness (Figure 2, B and C). Systolic performance was markedly impaired in ACC dHKO mice, with an approximately 51% reduction in EF and an approximately 62% reduction in FS at 10 months relative to WT (Figure 2C). These functional deficits were associated with increased expression of fetal cardiac genes Nppa, Nppb, and Myh7, molecular markers of pathological hypertrophy and heart failure (Figure 2D) (25, 26). Masson’s trichrome (TC) staining revealed extensive interstitial fibrosis, with a fibrotic area >5-fold greater in ACC dHKO hearts than in WT hearts (Figure 2E). Pulmonary congestion was also assessed in ACC dHKO mice. Lung fluid weight correlated with cardiac dysfunction (EF and FS) in ACC dHKO mice (Supplemental Figure 1A; supplemental material available online with this article; https://doi.org/10.1172/JCI202528DS1) (27), consistent with pulmonary edema accompanying progressive cardiac decompensation. However, lung weight/body weight ratios did not differ between WT and ACC dHKO mice (Supplemental Figure 1B).
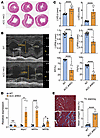 Figure 2
Figure 2Decreased heart function in ACC dHKO mice. (A) Transverse/2-chamber views of hearts from 11-month-old WT and ACC dHKO male mice. (B) Representative M-mode ECHO images from 10-month-old male WT and ACC dHKO mice. LVPW, left ventricular posterior wall. (C) ECHO assessment of cardiac function in 10-month-old WT and ACC dHKO male mice (n = 15 WT and 9 ACC dHKO). ****P < 0.0001 by unpaired 2-tailed Student’s t test. Data are presented as the mean ± SEM. (D) Total RNA was isolated from hearts of 10-week-old WT and ACC dHKO male mice, and RT-qPCR was performed to measure expression of hypertrophy-associated genes (Myh7, Nppa, and Nppb) (n = 5 WT and 5 ACC dHKO). *P < 0.05, ***P < 0.001 by unpaired 2-tailed Student’s t test. (E) Representative TC-stained sections from hearts of 11-month-old WT and ACC dHKO male mice are shown at ×10 magnification. Collagen area was quantified from TC-positive regions using multiple microscopy images from each group (11 images from 2 WT mice and 15 images from 3 ACC dHKO mice) with the split-red-channel method, as described in Methods. ****P < 0.0001 by unpaired 2-tailed Student’s t test.
Enhanced FAO in the heart has been implicated in impaired cardiac performance (2, 28). Earlier studies in ACC1- or ACC2-deficient mice demonstrated increased FAO specifically in the affected tissues, driven by reduced malonyl-CoA levels. This reduction relieves CPT1 inhibition and enhances mitochondrial FA import and β-oxidation (18, 21, 29–31). Based on these findings, we hypothesized that cardiomyocyte injury in ACC double-knockout (ACC dHKO) hearts results from excessive FAO. To test this, we first assessed CPT1 activity by quantifying long-chain acylcarnitines, products of CPT1-mediated mitochondrial FA transport. Long-chain acylcarnitines were elevated in ACC dHKO hearts, whereas octanoylcarnitine, which enters mitochondria independent of CPT1, was unchanged (Figure 3A).
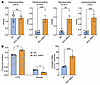 Figure 3
Figure 3Increased FAO in ACC dHKO hearts. (A) Fatty acylcarnitines were measured in hearts isolated from 24-week-old WT and ACC dHKO female mice, as described in Methods (n = 7 WT and 4 ACC dHKO). (B) Cardiac FA and glucose oxidation rates were measured with Langendorff working heart perfusion in live hearts isolated from 7-week-old WT or ACC dHKO mice (n = 4 per group), as described in Methods. LCFA, long-chain fatty acid. Data are presented as the mean ± SEM. *P < 0.05, ***P < 0.001, ****P < 0.0001 by unpaired 2-tailed Student’s t test.
Next, we examined whether FAO was elevated in ACC dHKO hearts due to enhanced CPT1 activity. FA and glucose oxidation rates were measured in isolated Langendorff-perfused WT and ACC dHKO hearts with [U-13C] long-chain FA and [1,6-13C2] glucose tracers. After a 30 min equilibration period to achieve metabolic steady state, fractional substrate oxidation was quantified. ACC dHKO hearts exhibited higher FAO and lower glucose oxidation, leading to an approximately 2-fold higher FA/glucose oxidation ratio than WT hearts (Figure 3B). These data indicate that ACC deficiency leads to increased CPT1-dependent FAO in the heart.
Given the established link between oxidative stress and mitochondrial dysfunction (32–34), we next assessed mitochondrial integrity in ACC dHKO hearts. Electron microscopy revealed wider cristae in cardiac mitochondria from ACC dHKO hearts than in WT hearts (Supplemental Figure 1, C and D). This crista dilation is consistent with mitochondrial ultrastructural changes reported in failing hearts and in experimental models of cardiac stress (35, 36). Despite this ultrastructural change, mitochondrial marker proteins and DNA copy number were unchanged between groups, indicating preserved overall mitochondrial content (Supplemental Figure 1, E and F).
Next, to assess overall mitochondrial function, mitochondria were isolated and mitochondrial respiratory function was measured with Seahorse (37, 38). ACC dHKO mitochondria exhibited approximately 53% lower maximal respiration with complex I substrates (pyruvate/malate) and approximately 32% lower respiration with complex II substrates (succinate + rotenone) than WT mitochondria (Figure 4A). In contrast, complex IV–driven respiration (antimycin/TMPD/ascorbate) was unchanged. These data indicate selective impairment of complexes I–III in ACC dHKO hearts.
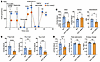 Figure 4
Figure 4Impaired respiratory electron transfer function in mitochondria isolated from ACC dHKO hearts. (A) Mitochondria were isolated from WT and ACC dHKO male mice aged 2–3 months (18 total mice; Seahorse experiments performed in 5 independent runs, 3–4 mice per run with age-matched WT and ACC dHKO in each run). Electron transfer between respiratory complexes was assessed by measuring oxygen consumption rate (OCR) during sequential additions of pyruvate/malate/FCCP, rotenone, succinate, antimycin A, and TMPD/ascorbate, as described in Methods. (B–D) Hearts from 20-week-old female WT and ACC dHKO mice were used (n = 6 WT and 7 ACC dHKO). (B) Free CoA and CoA thioesters (propionyl-CoA and succinyl-CoA) were quantified by LC-MS as described for malonyl-CoA and acetyl-CoA. (C) NAD, NADH, and the NAD+/NADH ratio were measured by LC-MS in freeze-clamped hearts. (D) Total adenine nucleotides levels were quantified by LC-MS. Cellular energy charge was calculated as ([ATP] + 0.5 × [ADP])/([ATP] + [ADP] + [AMP]). Data are presented as the mean ± SEM. *P < 0.05, ***P < 0.001, ****P < 0.0001 by unpaired 2-tailed Student’s t test.
To investigate the metabolic basis of this defect, we measured mitochondrial cofactors involved in TCA cycle flux. Free CoA levels were markedly reduced in ACC dHKO hearts, while acetyl-CoA remained unchanged (Figure 4B; see also Figure 1C). Succinyl-CoA was decreased, whereas propionyl-CoA showed a downward trend, consistent with CoA depletion limiting CoA-dependent TCA steps (39, 40).
NAD+ and ATP are generated during oxidative metabolism, and levels decline in failing hearts (41–43). NAD+ and ATP levels in ACC dHKO hearts were reduced by approximately 25% and approximately 14%, respectively (Figure 4, C and D). NADH levels remained unchanged, producing a more reduced NAD+/NADH ratio consistent with impaired electron transport. Despite lower ATP, total adenine nucleotides (ATP + ADP + AMP) and cellular energy charge were preserved, indicating maintenance of global adenine nucleotide homeostasis. Together, these findings demonstrate that ACC loss in cardiomyocytes leads to reduced essential mitochondrial cofactors, including free CoA and NAD+-constraining TCA cycle flux, redox balance, and oxidative phosphorylation capacity.
We next assessed whether excessive FAO alters cardiac FA composition. ACC dHKO hearts exhibited broad reductions in multiple FAs, including 16:0 (–24%), 18:1n9 (–56%), and 20:4n6 (–34%), with the largest decrease observed in linoleic acid (18:2n6; ~60%) (Supplemental Figure 2A).
To determine which lipid pools accounted for this loss, cardiac lipids were fractionated into neutral and phospholipid fractions. Neutral lipids (triacylglycerol, diacylglycerol, and cholesteryl esters) and their FA compositions were unchanged (Supplemental Figure 2, B–D). In contrast, phospholipids underwent marked remodeling (Figure 5). Although total phosphatidylethanolamine (PE) and phosphatidylglycerol (PG) were preserved (Figure 5, A and B), total phosphatidylcholine (PC) and phosphatidylinositol (PI) were reduced by 36% and 71%, respectively (Figure 5, C and D). Importantly, this decrease reflected a selective loss of linoleoyl-containing phospholipid species: most 18:2-containing phospholipids were markedly reduced in ACC dHKO hearts (Figure 5, A–D), indicating specific depletion of linoleoyl-phospholipids rather than a global reduction in phospholipid abundance.
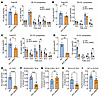 Figure 5
Figure 5Reduced linoleoyl phospholipids and tetralinoleoyl cardiolipin in ACC dHKO hearts. (A–E) Cardiac phospholipids from 8-week-old male WT and ACC dHKO mice were analyzed by LC-MS/MS, as described in Methods. Total phospholipids, each PL species, and their specific FA compositions were quantified (n = 5 WT and 5 ACC dHKO for A–C and E; n = 4 WT and 4 ACC dHKO for D). PE (A), PG (B), PC (C), PI (D), and linoleoyl cardiolipin (72:8, E). IS, internal standard. (F–I) Incorporation of labeled FAs into phospholipids and cardiolipins in WT and ACC dHKO hearts. Mice received intraperitoneal injections of LA-d4 (18:2) or OA-d17 (18:1). Enrichment of labeled species was determined 1 h after injection (n = 5 WT and 5 ACC dHKO). MS analysis quantified PC34:2 (16:0,18:2; D4-LA) (F), PC34:1 (16:0,18:1; D17-OA) (G), CL72:6 containing 1 18:2 chain (D4-LA) (H), and CL72:6 containing 1 18:1 chain (D17-OA) (I). Enrichment was calculated as labeled/(labeled + unlabeled) from isotopologue distributions, as described in Methods. Data are presented as the mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 by unpaired 2-tailed Student’s t test.
Inasmuch as linoleic acid is an essential FA that cannot be synthesized de novo, we next determined whether this reduction was due to impaired uptake into cardiomyocytes. To address this, we initially inhibited mitochondrial degradation of infused FAs in ACC dHKO mice by administering the CPT1 inhibitor etomoxir for 40 min and then infused [3H]-linoleic acid or [3H]-oleic acid for 10 min. The radioactivity was subsequently measured in organic and aqueous phases of heart extracts. No differences in the uptake of 18:2 or 18:1 were measured between WT and ACC dHKO hearts (Supplemental Figure 2, E and F).
The marked reduction of linoleic acid–containing phospholipids prompted us to investigate cardiolipin, a mitochondria-specific phospholipid that accounts for approximately 20% of total mitochondrial lipids and plays a key role in maintaining respiratory chain function. The predominant mature form of cardiolipin contains four linoleic acid residues (44, 45), and cardiolipin is essential for optimal electron transfer by stabilizing respiratory supercomplexes and supporting efficient oxidative phosphorylation (46–51). Given the close association between cardiolipin composition and mitochondrial respiration as well as the abundance of linoleic acid in mature cardiolipin, we measured cardiolipin species in ACC dHKO hearts. Figure 5E shows that tetralinoleoyl cardiolipin (CL72:8) was approximately 80% lower in ACC dHKO hearts than in WT controls, indicating a marked loss of mature cardiolipin.
Cardiolipin biosynthesis involves 2 main steps: the generation of a premature cardiolipin from PG by cardiolipin synthase, followed by remodeling into the mature form by tafazzin, an acyltransferase that incorporates linoleic acid from 18:2-PC or 18:2-PE (44, 45, 52–54). RT-qPCR analysis revealed that cardiolipin synthase and tafazzin mRNA levels were unchanged in ACC dHKO hearts (Supplemental Figure 2G). In line with this, PG levels were also unaffected (Figure 5B). In contrast, both 18:2-PC and 18:2-PE, which serve as acyl donors for cardiolipin maturation, were markedly decreased by 56% and 55%, respectively, in ACC dHKO hearts (Figure 5, A and C).
To determine whether the observed differences in levels of 18:2-containing phospholipids and the mature cardiolipin in ACC dHKO hearts were a result of diminished synthesis, we performed an in vivo synthesis assay with isotope-labeled FAs. WT and ACC dHKO mice were administered linoleic acid-d4 (LA-d4, 18:2) dissolved in saline via intraperitoneal injection. For comparison, oleic acid-d17 (OA-d17, 18:1) was also injected. One hour later, incorporation of LA-d4 into newly synthesized PC34:2 was reduced by 62% in ACC dHKO hearts relative to WT (Figure 5F), whereas incorporation of OA-d17 into PC34:1 was unchanged (Figure 5G). Cardiolipin labeling mirrored this pattern: LA-d4 incorporation into cardiolipin was reduced by 53% in ACC dHKO hearts, whereas OA-d17 incorporation did not differ between groups (Figure 5, H and I). These results indicate that the marked loss of tetralinoleoyl CL (CL72:8) in ACC dHKO hearts is primarily due to insufficient availability of linoleoyl-phospholipid precursors rather than impaired PG supply or reduced expression of cardiolipin biosynthetic enzymes.
Because mitochondrial dysfunction can promote oxidant release, we next assessed oxidative stress. Basal mitochondrial H2O2 release was slightly elevated in ACC dHKO hearts, though absolute values were low (Supplemental Figure 2H). However, when energized with succinate to drive complex I reverse electron transport, ACC dHKO mitochondria produced markedly less H2O2 than WT (Supplemental Figure 2I), consistent with reduced electron transport capacity. In line with this, thiobarbituric acid reactive substance (TBARS) analysis revealed no increase in lipid peroxidation (Supplemental Figure 2J), and protein carbonylation exhibited only a modest, nonsignificant trend toward elevation (Supplemental Figure 2K). Thus, ACC dHKO hearts show no evidence of increased oxidative injury, and reverse electron transport–dependent mitochondrial ROS production is in fact reduced.
PPARα serves as a critical regulator of FAO in the heart, and its activity is reduced proportionally with the decrease of FAO in both animal models and humans with cardiac hypertrophy (55, 56). Downregulation of PPARα is considered the primary mechanism responsible for the shift in substrate utilization from FAs to glucose during the progression of heart failure (2). In hearts of ACC dHKO mice, PPARα signaling was suppressed, similar to findings previously reported in livers of ACC hepatocyte-specific knockout mice (18). In Supplemental Figure 3A, mRNA levels of PPARα-dependent genes were 20%–30% lower in hearts from 14-week-old ACC dHKO mice (2 left bars for each gene).
To determine whether restoring PPARα activity normalizes the expression of PPARα-regulated genes, the PPAR agonist WY-14643 was administered to 8-week-old ACC dHKO mice for 6 weeks. In Supplemental Figure 3A, WY-14643 restored expression of PPARα-induced genes (Cpt1a, Cpt1b, Cpt2, HmgCS2, and Mcd) to levels observed in untreated WT hearts. However, despite normalization of PPARα-dependent gene expression, WY-14643 treatment did not improve cardiac function in ACC dHKO mice (Supplemental Figure 3B). Remarkably, even early intervention was detrimental: ACC dHKO mice treated with WY-14643 starting at 4 weeks of age exhibited worse cardiac function after 4 weeks of treatment than those treated beginning at 8 weeks (Supplemental Figure 3C).
To determine whether excessive FAO directly drives the loss of cardiolipin and subsequent heart failure in ACC dHKO hearts, we inhibited FAO with etomoxir. Etomoxir is widely employed in animal studies or cultured cells as an inhibitor of FAO (57, 58). WT and ACC dHKO mice were fed chow supplemented with etomoxir (20 mg/kg/day) for 1 month, starting at 4 weeks of age. FA composition and cardiolipin levels in the hearts of WT and ACC dHKO mice were then measured. As shown in Figure 6A, etomoxir increased total C18 FAs in the heart, with the largest increase observed in linoleic acid in both WT and ACC dHKO mice.
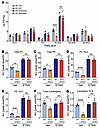 Figure 6
Figure 6Inhibition of FAO by etomoxir normalizes the amount of linoleic acid in ACC dHKO hearts. (A) Hearts from 4-week-old WT and ACC dHKO male mice fed chow or chow supplemented with etomoxir (20 mg/kg/day) for 1 month were harvested, and FA composition was measured by gas chromatography, as described in Methods (n = 4 WT-NT, 4 ACC dHKO-NT, 5 WT-etomoxir, and 7 ACC dHKO-etomoxir). (B–G) LC-MS/MS was used to quantify total phospholipids (B, PC; C, PE), linoleoyl-containing phospholipids (D, PC 18:2; E, PE 18:2), total cardiolipins (F), and linoleoyl cardiolipins (G, CL 72:8), in hearts from A, as described in Methods (n = 4 WT-NT, 4 ACC dHKO-NT, 5 WT-etomoxir, and 6 ACC dHKO-etomoxir). Data are presented as the mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001 by unpaired 2-tailed Student’s t test.
We next analyzed phospholipid species by liquid chromatography–tandem mass spectrometry (LC-MS/MS). Etomoxir increased PC and PE by approximately 2-fold in both genotypes (Figure 6, B and C) and markedly increased linoleoyl-PC and linoleoyl-PE by approximately 3-fold and approximately 10-fold, respectively (Figure 6, D and E). Importantly, this treatment equalized linoleoyl-phospholipid levels between WT and ACC dHKO hearts. Interestingly, total and mature cardiolipin levels were unchanged in WT hearts with etomoxir, but both were restored to WT levels in ACC dHKO hearts (Figure 6, F and G). The normalization of linoleic acid concentrations and cardiolipin levels in ACC dHKO hearts following the inhibition of FAO by etomoxir suggests that these changes are primarily driven by excessive FAO.
We next investigated whether etomoxir administration also restores the normal flow of mitochondrial electron transfer through respiratory complexes in the hearts of ACC dHKO mice. WT and ACC dHKO mice, 8 weeks of age, were fed chow supplemented with etomoxir (20 mg/kg/day) for 2 weeks. The mice were euthanized for cardiac mitochondria isolation, and the function of electron transport was measured as described in Figure 7A. Oxygen consumption rates were similar across all mitochondrial complexes in mitochondria from WT and ACC dHKO hearts after etomoxir treatment, suggesting that restored cardiolipin levels normalize mitochondrial electron flow in ACC dHKO hearts.
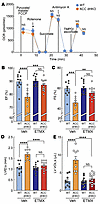 Figure 7
Figure 7Inhibition of FAO by etomoxir prevents the development of heart failure in ACC dHKO mice. (A) Eight-week-old WT and ACC dHKO male mice were fed either chow or chow containing etomoxir (20 mg/kg/day) for 2 weeks. Heart mitochondria were isolated, and oxygen consumption rates (OCRs) were measured during sequential additions of pyruvate/malate/FCCP, rotenone, succinate, antimycin A, and TMPD/ascorbate, as described in Methods (n = 10 WT and 10 ACC dHKO). Data are presented as the mean ± SEM; comparisons were made by unpaired 2-tailed Student’s t test. (B–E) Four-week-old WT and ACC dHKO male mice were fed chow or chow supplemented with etomoxir (20 mg/kg/day) for 1 month. ECHO was performed to quantify EF (B), FS (C), LVIDs (D), and LV Vols (E) (n = 11 WT-NT, 9 ACC dHKO-NT, 12 WT-etomoxir, and 13 ACC dHKO-etomoxir for B and C; n = 10 WT-NT, 9 ACC dHKO-NT, 12 WT-etomoxir, and 13 ACC dHKO-etomoxir for D and E). Data are presented as the mean ± SEM. **P < 0.01, ***P < 0.001 by 2-way ANOVA (genotype × treatment) with Šidák’s multiple-comparison test.
Finally, we assessed whether blocking FAO with etomoxir improves the cardiac function of ACC dHKO mice. Mice for these studies are those described for the cardiolipin measurements described above. Chow supplemented with etomoxir (20 mg/kg/day) was fed to 4-week-old WT and ACC dHKO mice for 4 weeks. It is important to note that ACC dHKO mice at 4 weeks of age do not exhibit evidence of heart failure (Supplemental Figure 4, A and B). As controls, another group of WT and ACC dHKO mice of the same age were fed chow diet. Over the course of 1 month, chow diet–fed ACC dHKO mice developed statistically significant cardiac dysfunction (Figure 7, B–E). Specifically, the EF and FS were reduced by 16% and 28%, respectively, and LVIDs were enlarged by 50% in ACC dHKO hearts. In contrast, hearts from ACC dHKO mice treated with etomoxir displayed normal EF, FS, and LVIDs (Figure 7, B–D). These results demonstrate that suppressing FAO with etomoxir effectively prevents the development of heart failure in ACC dHKO mice.
-
Discussion
In this study, we demonstrate that loss of ACC1 and ACC2 in cardiomyocytes results in unchecked FAO, depletion of linoleic acid–containing phospholipids, severe cardiolipin deficiency, mitochondrial respiratory failure, and ultimately dilated cardiomyopathy. These findings establish that balanced ACC activity and malonyl-CoA signaling are essential to maintain cardiac lipid composition, mitochondrial function, and contractile performance.
The healthy heart flexibly oxidizes FAs and glucose depending on workload, substrate availability, and hormonal state (28, 59–61). In contrast, failing hearts progressively lose this metabolic flexibility and shift toward greater reliance on glucose oxidation (2, 62). Although the role of FAO in heart failure has been debated, multiple proof-of-concept clinical studies suggest that enhancing glucose oxidation can improve cardiac function. This metabolic shift naturally involves reduced FAO through the Randle cycle (28, 63–67). Consistent with this, direct inhibition of FAO through CPT1 blockade improves cardiac function in myocardial infarction (58), ischemia–reperfusion (68), and pacing-induced heart failure (57) models, and a pilot trial in patients also reported benefit (16). In agreement with these data, etomoxir treatment fully prevented heart failure in ACC dHKO mice, while oxfenicine, a second compound that inhibits CPT1, produced similar protection and normalized cardiac lipid composition (Figure 7 and Supplemental Figure 4, C–G). However, initiating etomoxir treatment after cardiac dysfunction developed (~20 weeks) did not improve function (Supplemental Figure 5, A–F), suggesting that reducing FAO is protective early but does not reverse established disease.
Kolwicz (29) reported that cardiomyocyte-specific deletion of ACC2 improved energetic efficiency and protected against pressure overload–induced dysfunction. Their model reduced malonyl-CoA by approximately 50% and modestly increased FAO — conditions that appear adaptive under stress. In contrast, the near-complete loss of malonyl-CoA in our ACC dHKO hearts produced sustained FAO hyperactivation sufficient to cause cardiomyopathy at baseline. Thus, the degree and context of FAO activation determine whether the metabolic shift is beneficial or harmful. Moreover, although Myh6-Cre mice can develop late-onset cardiac dysfunction (69, 70), 3 observations demonstrate that the phenotype here is not due to Cre toxicity: (a) graded severity from ACC1 HKO < ACC2 HKO < ACC dHKO indicates a gene dosage effect; (b) etomoxir completely normalized cardiac structure and function, which would not occur with Cre-mediated toxicity; and (c) abnormalities appeared early in ACC dHKO mice, whereas Myh6-Cre effects typically manifest at an older age (70, 71).
Mechanistically, loss of ACC increases FAO and depletes linoleic acid–containing phospholipid precursors, including PC and PE, which are required for the generation of mature tetralinoleoyl cardiolipin (Figure 5, A, C, and F–I). In this context, the preferential reduction of multiple FA species in phospholipids — despite preserved levels in neutral lipids such as triacylglycerol, diacylglycerol, and cholesteryl esters — suggests a selective vulnerability of membrane lipids to altered FA flux. Because the heart relies predominantly on lipid oxidation for energy and has limited capacity to store lipids, neutral lipid pools often remain relatively stable even when FA availability or utilization is perturbed (59, 72, 73). By contrast, phospholipids, particularly those comprising mitochondrial membranes, are more sensitive to shifts in FA supply (74). Notably, 18:2 linoleic acid, the dominant acyl chain in cardiolipin, was the most strongly reduced in ACC dHKO hearts, supporting the concept that unrestrained FAO constrains cardiolipin remodeling by limiting linoleate-containing precursor pools.
Given the central role of cardiolipin in organizing the respiratory chain and supporting mitochondrial function, a link between impaired cardiolipin remodeling and mitochondrial dysfunction is well established in Barth syndrome. Cardiolipin is a mitochondrial inner membrane phospholipid that is essential for normal respiratory chain activity (44, 46), and loss of tafazzin — the cardiolipin remodeling enzyme mutated in Barth syndrome — lowers cardiolipin and causes dilated cardiomyopathy in humans as well as in tafazzin-deficient mouse models (75, 76). In these models, cardiolipin deficiency selectively impairs complex I–III function, whereas complex IV activity is relatively preserved (75, 77, 78). Consistent with this paradigm, respiration in mitochondria isolated from ACC dHKO hearts exhibited a pattern similar to that reported in TAZ knockdown or knockout cells (Figure 4A). Together, these observations support cardiolipin deficiency as a major driver of respiratory chain impairment and cardiomyopathy in ACC dHKO hearts, underscoring the requirement for cardiolipin in maintaining complex I–III activity and supercomplex integrity.
Given that disruptions in cardiolipin and respiratory chain organization can secondarily increase ETC-derived oxidative stress, we evaluated whether oxidative damage accompanies cardiolipin loss in ACC dHKO hearts. However, succinate-driven H2O2 release (reflecting reverse electron transport at complex I) was reduced relative to WT, TBARS levels were unchanged, and the trend toward increased protein carbonylation did not reach statistical significance (Supplemental Figure 2, H–K). These findings argue that cardiolipin loss in this model primarily compromises electron transfer efficiency and metabolic balance rather than promoting a generalized increase in oxidative stress.
This framework also helps interpret the stage-dependent shifts in substrate utilization observed in ACC dHKO hearts. The healthy heart adjusts its relative use of FAs and glucose according to workload, substrate availability, and hormonal state, whereas heart failure is often accompanied by progressive loss of metabolic flexibility and a relative shift toward glucose oxidation (2, 28, 62, 79). In ACC dHKO hearts, the FA/glucose oxidation ratio was elevated at 7 weeks but markedly reduced at 10 months, when heart failure was established (Figure 3 and Supplemental Figure 6, A and B). Although reduced FAO at advanced stages of heart failure has been proposed to limit oxygen consumption and oxidative stress and thereby confer functional protection (61, 80–82), an actively regulated metabolic switch is unlikely to account for the late-stage decline in ACC dHKO hearts because malonyl-CoA–mediated control of CPT1 is genetically ablated. Instead, the reduction in FAO appears to be more consistent with a secondary loss of mitochondrial oxidative capacity arising from cardiolipin deficiency and respiratory chain dysfunction.
The selective depletion of linoleate-containing phospholipids in ACC dHKO hearts further raises the question of whether linoleate is lost through oxidative degradation, such as ROS-driven lipid peroxidation, or preferential consumption under sustained FAO. The absence of elevated TBARS and other oxidative damage markers argues against lipid peroxidation as a dominant mechanism. Rather, the data are consistent with a model in which persistently elevated CPT1-dependent FAO promotes preferential β-oxidation of linoleate, thereby depleting linoleoyl-PC/PE pools required for cardiolipin remodeling.
This interpretation is also consistent with the limited efficacy of dietary linoleic acid supplementation. Sunflower oil feeding increased cardiac linoleic acid (C18:2n6) to a similar absolute extent in WT and ACC dHKO hearts (Supplemental Figure 6D), but because baseline linoleate levels were markedly lower in ACC dHKO hearts, this increase was insufficient to restore steady-state linoleate levels and cardiac dysfunction persisted (Supplemental Figure 6E). Thus, under conditions of persistently elevated FAO, increased dietary linoleate can raise cardiac linoleate content but may not achieve the steady-state levels required to support normal phospholipid remodeling, providing a mechanistic explanation for the failure of dietary linoleic acid supplementation to rescue cardiac dysfunction in ACC dHKO hearts.
Along with cardiolipin deficiency, another contributing factor to mitochondrial dysfunction in ACC dHKO hearts may be impaired glucose oxidation. As demonstrated by Elnwasany et al. (83), ACC is required for insulin-stimulated activation of pyruvate dehydrogenase (PDH) via malonyl-CoA–mediated inhibition of FAO. In the absence of ACC, elevated β-oxidation suppresses PDH activity through sustained phosphorylation, leading to decreased glucose oxidation via the TCA cycle. Therefore, the mitochondrial dysfunction observed in ACC dHKO hearts may be compounded by both a deficiency in cardiolipin-dependent electron transport and an impairment in PDH-driven glucose oxidation, further limiting mitochondrial ATP production capacity.
An unexpected observation was the increased docosahexaenoic acid (DHA) levels in ACC dHKO hearts (Supplemental Figure 2A). Sullivan et al. (84) reported elevated cardiac DHA in diabetic humans, and DHA supplementation in mice leads to cardiolipin remodeling with increased DHA incorporation, accompanied by reduced respiratory complex activity. Consistent with this, DHA-containing cardiolipins (CL76:12, CL76:11, and CL76:10) were markedly elevated in ACC dHKO hearts (~5% in WT vs. ~20% in ACC dHKO; Supplemental Figure 7, A and B). Notably, CPT1 inhibition with etomoxir reduced DHA-containing cardiolipin in both WT and ACC dHKO hearts and eliminated the genotype difference (Supplemental Figure 7C), indicating that this remodeling is FAO dependent. Although we cannot determine from these data whether DHA-containing cardiolipin directly contributes to the decline in cardiac function, its FAO-sensitive regulation and parallel reduction with functional recovery highlight this axis as a potential contributor and a priority for future mechanistic studies.
In addition, analysis of canonical “immature” CL isoforms supports the conclusion that the loss of mature tetralinoleoyl CL (CL72:8) in ACC dHKO hearts reflects impaired maturation/remodeling rather than reduced synthesis of immature CL. These immature species (CL70:X), which serve as tafazzin-remodeling substrates (85, 86), were not decreased in ACC dHKO hearts and, in some cases, were modestly increased (Supplemental Figure 7D). Thus, these data support our interpretation that reduced mature CL in ACC dHKO hearts is not driven by diminished immature CL synthesis, but instead reflects a defect in the maturation/remodeling step due to limited availability of linoleate-containing PC and PE.
In summary, cardiomyocyte-specific loss of ACC1 and ACC2 eliminates malonyl-CoA, leading to uncontrolled CPT1-dependent FAO. This sustained FAO depletes tetralinoleoyl cardiolipin — the dominant and functionally essential mitochondrial cardiolipin species — resulting in impaired electron transport, reduced ATP production, and the development of heart failure in ACC dHKO mice. Thus, sustained FAO hyperactivation in this model produces profound energetic and mitochondrial failure, yielding a state of metabolic inefficiency that is conceptually consistent with the broader framework of the failing heart as “an engine running out of fuel (2).” Given the rising burden of cardiomyopathy and limitations of current therapies, our findings highlight the importance of ACC-driven malonyl-CoA signaling and cardiolipin homeostasis in cardiac energy metabolism. Targeting FAO and cardiolipin pathways may offer new therapeutic strategies to prevent or treat heart failure.
-
Methods
Sex as a biological variable. ECHO was performed in male mice because imaging stability and probe positioning were more consistent in males. Biochemical endpoints (including lipidomics, ATP/ADP, and redox assays) were measured in both sexes, and no sex-dependent differences were observed. The study was not powered to detect small sex effects beyond these assays.
Animals. Acc1-floxed alleles were generated as we previously described (18). Acc2fl/fl mice were purchased from The Jackson Laboratory (stock 013042; B6N;129S-Acacbtm1.1Lowl/J). ACC1 and ACC2 cardiomyocyte-specific double knockout mice were generated by crossing Acc1fl/fl and Acc2fl/fl mice with Myh6-Cre transgenic mice obtained from The Jackson Laboratory [stock 011038; B6.FVB-Tg(Myh6-cre)2182Mds/J]. Mice were housed at room temperature (23°C) and maintained on a 12 h light/12 h dark cycle and provided access to rodent chow diet ad libitum (Harlan, Teklad Global 18% Protein Rodent Diet 2018; 18% kcal from fat, 3.1 kcal/g). Etomoxir was purchased from Adooq Bioscience and fed ad libitum (20 mg/kg/day) after mixing into the powdered chow diet. For ECHO studies, control flox/flox mice and ACC dHKO mice were fed an etomoxir-supplemented diet (20 mg/kg/day) for 1 month, and for Seahorse studies, mice were fed the etomoxir supplemented diet for 2 weeks prior to study. A detailed experimental design is provided in Supplemental Methods.
RNA extraction and qPCR. RNA was isolated with RNA-Stat60, treated with DNase, and reverse-transcribed with TaqMan reagents (Applied Biosystems, Thermo Fisher Scientific). qPCR was performed as described (87). Primer sequences are provided in the supplemental materials.
ECHO (conscious). Cardiac function was evaluated by transthoracic ECHO with a VisualSonics Vevo 2100 system equipped with an MS400C probe (FUJIFILM VisualSonics) (88). M-mode images at the papillary muscle level were acquired, and EF, FS, and LV dimensions as well as wall thickness were calculated per standard guidelines.
Langendorff heart perfusions. Ex vivo substrate utilization was measured in Langendorff-perfused mouse hearts supplied with 13C-labeled glucose and FAs. 13C-NMR spectra were collected and analyzed by isotopomer modeling to quantify fractional oxidation of each substrate. Experimental conditions, tracers, and analysis parameters are described in Supplemental Methods.
FA composition measurements. Lipids were extracted from mouse hearts with chloroform:methanol (2:1) and dried under nitrogen gas. FA composition was measured with gas chromatography as described previously (89).
LC-MS/MS lipidomics. Lipids were extracted from approximately 50 mg heart tissue with dichloromethane:methanol-based liquid–liquid extraction with internal standards (Splash Lipidomix, Avanti). Samples were analyzed by LC-MS/MS (QTRAP 6500+), and lipid species were quantified by multiple reaction monitoring (MRM) and normalized to internal standards. Detailed LC-MS/MS parameters, extraction procedures, and MRM transitions are provided in Supplemental Methods.
Mitochondrial respiration. Isolated cardiac mitochondria (5 μg/well) were analyzed with a Seahorse XF24 analyzer (Agilent Technologies). Substrates and inhibitors were injected sequentially (pyruvate/malate/FCCP, rotenone, succinate, antimycin A, TMPD/ascorbate) to assess electron flow across complexes I–IV. Detailed mitochondrial isolation procedures, assay conditions, titration parameters, and sequential injection settings are provided in Supplemental Methods.
Short-chain acyl-CoA measurements. Frozen hearts were extracted in 10% trichloroacetic acid spiked with 13C-acetyl-CoA and 13C-malonyl-CoA, purified by solid-phase extraction, and analyzed by LC-MS/MS in MRM mode. Detailed extraction, solid-phase extraction conditions, instrument settings, and quantification parameters are provided in Supplemental Methods.
Fatty acyl carnitine measurements. Heart tissue was homogenized, spiked with deuterated internal standards, derivatized, separated by reverse-phase HPLC, and analyzed on a triple-quadrupole mass spectrometer (90, 91). Derivatization chemistry, LC gradient, MS transitions, and normalization strategy are detailed in Supplemental Methods.
In vivo cardiolipin synthesis assays. Mice received intraperitoneal LA-d4 and OA-d17. Hearts were collected 1 h later, lipids were extracted, and LC-MS/MS was used to quantify labeled and unlabeled PC species and cardiolipin. Fractional labeling was calculated as labeled/(labeled + unlabeled). Injection protocol, extraction solvent ratios, MS settings, and isotopologue calculation details are provided in Supplemental Methods.
FA uptake assays. Mice were pretreated with etomoxir (40 mg/kg) for 40 min prior to injection of [3H]-linoleic acid or [3H]-oleic acid. Hearts were collected 10 min later, extracted by the Folch method, and scintillation counting was used to measure radiolabel incorporation. Tracer preparation, sample handling, phase-separation workflow, and normalization procedures are described in Supplemental Methods.
Histology. Heart tissue sections were fixed in 10% (v/v) neutral buffered formalin. Paraffin embedding, sectioning, as well as H&E and TC staining were performed by the University of Texas Southwestern Medical Center’s Molecular Pathology Core. Mean areas of TC-stained collagen in each heart section were determined with standard ImageJ (NIH) software. TC-stained heart sections demonstrated blue collagen under light microscopy (bright field). The images were separated into 3 distinct channels. Collagen in heart sections was quantified by image reduction to red channel followed by automatic calculation of mean red area (minimum = 0; maximum = 255).
Statistics. Statistical analyses were performed with GraphPad Prism 10. Data are presented as mean ± SEM. Comparisons between 2 groups were performed with unpaired 2-tailed Student’s t test. For comparisons among multiple groups, 1-way ANOVA followed by Tukey’s post hoc test was used. P < 0.05 was considered statistically significant. The number of biological replicates (n) for each experiment is reported in figure legends. No statistical methods were used to predetermine sample size.
Study approval. All animal experiments were performed in accordance with and approved by the IACUC at the University of Texas Southwestern Medical Center. No human subjects were included in this study.
Data availability. Raw data underlying all graphs and reported values presented in the main text and supplemental figures are provided in the Supporting Data Values file. Lipidomics, LC-MS/MS, isotopic tracing, and Seahorse data are available from the corresponding author upon reasonable request. No high-throughput sequencing or microarray datasets were generated. All materials used in this study are listed in the supplemental materials.
-
Author contributions
CWK and JDH conceptualized the study and wrote the manuscript. CWK, GV, XF, JGM, CD, CL, GS, SD, and MAM performed investigation. JDH acquired funding. JDH, ZVW, CK, CRM, and SCB supervised the study. CWK, CK, and SD analyzed data. CWK, GV, XF, ZVW, CK, SCB, MAM, and JDH reviewed and edited the manuscript.
-
Conflict of interest
The authors have declared that no conflict of interest exists.
-
Funding support
This work is the result of NIH funding, in whole or in part, and is subject to the NIH Public Access Policy. Through acceptance of this federal funding, the NIH has been given a right to make the work publicly available in PubMed Central.
- NIH: P01HL160487 and P30DK127984 (to JDH).
- American Heart Association: 23SCEFIA1154964, 24IPA1272385, and 24TPA1297929 (to GS).
-
Acknowledgments
We thank Norma Anderson, Judy Sanchez, Tuyet Dang, Tam Tran, and Tessa Edwards for excellent technical assistance and Chelsea Burroughs and Nancy Heard for assistance with graphics.
Address correspondence to: Jay D. Horton, Center for Human Nutrition, Departments of Internal Medicine and Molecular Genetics, University of Texas Southwestern Medical Center, 5323 Harry Hines Blvd., Dallas, Texas 75390-9046, USA. Phone: 214.648.9677; Email: jay.horton@UTsouthwestern.edu.
-
Footnotes
Copyright: © 2026, Kim et al. This is an open access article published under the terms of the Creative Commons Attribution 4.0 International License.
Reference information: J Clin Invest. 2026;136(9):e202528. https://doi.org/10.1172/JCI202528.
See the related Commentary at Running rich: how excess fatty acid oxidation drains the cardiac engine.
-
References
- Doenst T, et al. Cardiac metabolism in heart failure: implications beyond ATP production. Circ Res. 2013;113(6):709–724.
- Neubauer S. The failing heart--an engine out of fuel. N Engl J Med. 2007;356(11):1140–1151.
- Ciarambino T, et al. Cardiomyopathies: an overview. Int J Mol Sci. 2021;22(14):7722.
- Virani SS, et al. Heart disease and stroke statistics-2020 update: a report from the American Heart Association. Circulation. 2020;141(9):e139–e596.
- Fillmore N, et al. Mitochondrial fatty acid oxidation alterations in heart failure, ischaemic heart disease and diabetic cardiomyopathy. Br J Pharmacol. 2014;171(8):2080–2090.
- Drazner MH. The progression of hypertensive heart disease. Circulation. 2011;123(3):327–334.
- Saliba LJ, Maffett S. Hypertensive heart disease and obesity: a review. Heart Fail Clin. 2019;15(4):509–517.
- Joseph JJ, et al. Comprehensive management of cardiovascular risk factors for adults with type 2 diabetes: a scientific statement from the american heart association. Circulation. 2022;145(9):722–759.
- Bornstein MR, et al. Human cardiac metabolism. Cell Metab. 2024;36(7):1456–1481.
- Lopaschuk GD, et al. Cardiac energy metabolism in heart failure. Circ Res. 2021;128(10):1487–1513.
- Aon MA, et al. Protective mechanisms of mitochondria and heart function in diabetes. Antioxid Redox Signal. 2015;22(17):1563–1586.
- Ritterhoff J, Tian R. Metabolism in cardiomyopathy: every substrate matters. Cardiovasc Res. 2017;113(4):411–421.
- Taegtmeyer H, Lubrano G. Rethinking cardiac metabolism: metabolic cycles to refuel and rebuild the failing heart. F1000Prime Rep. 2014;6:90.
- Glatz JFC, et al. Re-balancing cellular energy substrate metabolism to mend the failing heart. Biochim Biophys Acta Mol Basis Dis. 2020;1866(5):165579.
- Holubarsch CJ, et al. A double-blind randomized multicentre clinical trial to evaluate the efficacy and safety of two doses of etomoxir in comparison with placebo in patients with moderate congestive heart failure: the ERGO (etomoxir for the recovery of glucose oxidation) study. Clin Sci (Lond). 2007;113(4):205–212.
- Schmidt-Schweda S, Holubarsch C. First clinical trial with etomoxir in patients with chronic congestive heart failure. Clin Sci (Lond). 2000;99(1):27–35.
- Abu-Elheiga L, et al. Human acetyl-CoA carboxylase: characterization, molecular cloning, and evidence for two isoforms. Proc Natl Acad Sci U S A. 1995;92(9):4011–4015.
- Kim CW, et al. Acetyl CoA carboxylase inhibition reduces hepatic steatosis but elevates plasma triglycerides in mice and humans: a bedside to bench investigation. Cell Metab. 2017;26(2):394–406.
- McGarry JD, et al. A possible role for malonyl-CoA in the regulation of hepatic fatty acid oxidation and ketogenesis. J Clin Invest. 1977;60(1):265–270.
- Abu-Elheiga L, et al. The subcellular localization of acetyl-CoA carboxylase 2. Proc Natl Acad Sci U S A. 2000;97(4):1444–1449.
- Essop MF, et al. Reduced heart size and increased myocardial fuel substrate oxidation in ACC2 mutant mice. Am J Physiol Heart Circ Physiol. 2008;295(1):H256–H265.
- Abu-Elheiga L, et al. Continuous fatty acid oxidation and reduced fat storage in mice lacking acetyl-CoA carboxylase 2. Science. 2001;291(5513):2613–2616.
- Glund S, et al. Inhibition of acetyl-CoA carboxylase 2 enhances skeletal muscle fatty acid oxidation and improves whole-body glucose homeostasis in db/db mice. Diabetologia. 2012;55(7):2044–2053.
- Boone AN, et al. Multiple-site phosphorylation of the 280 kDa isoform of acetyl-CoA carboxylase in rat cardiac myocytes: evidence that cAMP-dependent protein kinase mediates effects of beta-adrenergic stimulation. Biochem J. 1999;341(pt 2):347–354.
- Li C, et al. IRE1α mediates the hypertrophic growth of cardiomyocytes through facilitating the formation of initiation complex to promote the translation of TOP-motif transcripts. Circulation. 2024;150(13):1010–1029.
- Dai C, et al. Lactate dehydrogenase a governs cardiac hypertrophic growth in response to hemodynamic stress. Cell Rep. 2020;32(9):108087.
- Ramírez A, Abelmann WH. Cardiac decompensation. N Engl J Med. 1974;290(9):499–501.
- Stanley WC, et al. Myocardial substrate metabolism in the normal and failing heart. Physiol Rev. 2005;85(3):1093–1129.
- Kolwicz SC, JrCardiac-specific deletion of acetyl CoA carboxylase 2 prevents metabolic remodeling during pressure-overload hypertrophy. Circ Res. 2012;111(6):728–738.
- Choi CS, et al. Continuous fat oxidation in acetyl-CoA carboxylase 2 knockout mice increases total energy expenditure, reduces fat mass, and improves insulin sensitivity. Proc Natl Acad Sci U S A. 2007;104(42):16480–16485.
- Abu-Elheiga L, et al. Acetyl-CoA carboxylase 2-/- mutant mice are protected against fatty liver under high-fat, high-carbohydrate dietary and de novo lipogenic conditions. J Biol Chem. 2012;287(15):12578–12588.
- Guo C, et al. Oxidative stress, mitochondrial damage and neurodegenerative diseases. Neural Regen Res. 2013;8(21):2003–2014.
- Quijano C, et al. Interplay between oxidant species and energy metabolism. Redox Biol. 2016;8:28–42.
- Ly LD, et al. Oxidative stress and calcium dysregulation by palmitate in type 2 diabetes. Exp Mol Med. 2017;49(2):e291.
- Mahata SK, et al. Effect of heart failure on catecholamine granule morphology and storage in chromaffin cells. J Endocrinol. 2016;230(3):309–323.
- Zhang J, et al. Mitochondrial phosphatase PTPMT1 is essential for cardiolipin biosynthesis. Cell Metab. 2011;13(6):690–700.
- An YA, et al. Dysregulation of amyloid precursor protein impairs adipose tissue mitochondrial function and promotes obesity. Nat Metab. 2019;1(12):1243–1257.
- Crewe C, et al. Extracellular vesicle-based interorgan transport of mitochondria from energetically stressed adipocytes. Cell Metab. 2021;33(9):1853–1868.
- Koves TR, et al. Pyruvate-supported flux through medium-chain ketothiolase promotes mitochondrial lipid tolerance in cardiac and skeletal muscles. Cell Metab. 2023;35(6):1038–1056.
- Wang Y, et al. Propionate-induced changes in cardiac metabolism, notably CoA trapping, are not altered by l-carnitine. Am J Physiol Endocrinol Metab. 2018;315(4):622–633.
- Cantó C, et al. NAD(+) metabolism and the control of energy homeostasis: a balancing act between mitochondria and the nucleus. Cell Metab. 2015;22(1):31–53.
- Tong D, et al. NAD+ repletion reverses heart failure with preserved ejection fraction. Circ Res. 2021;128(11):1629–1641.
- Abdellatif M, et al. NAD+ metabolism in cardiac health, aging, and disease. Circulation. 2021;144(22):1795–1817.
- Dudek J, et al. The role of mitochondrial cardiolipin in heart function and its implication in cardiac disease. Biochim Biophys Acta Mol Basis Dis. 2019;1865(4):810–821.
- Sparagna GC, Lesnefsky EJ. Cardiolipin remodeling in the heart. J Cardiovasc Pharmacol. 2009;53(4):290–301.
- Dolinsky VW, et al. Cardiac mitochondrial energy metabolism in heart failure: role of cardiolipin and sirtuins. Biochim Biophys Acta. 2016;1861(10):1544–1554.
- Raval KK, Kamp TJ. Cardiomyopathy, mitochondria and Barth syndrome: iPSCs reveal a connection. Nat Med. 2014;20(6):585–586.
- Mulligan CM, et al. Dietary linoleate preserves cardiolipin and attenuates mitochondrial dysfunction in the failing rat heart. Cardiovasc Res. 2012;94(3):460–468.
- Farvid MS, et al. Dietary linoleic acid and risk of coronary heart disease: a systematic review and meta-analysis of prospective cohort studies. Circulation. 2014;130(18):1568–1578.
- Robinson NC. Functional binding of cardiolipin to cytochrome c oxidase. J Bioenerg Biomembr. 1993;25(2):153–163.
- Maekawa S, et al. Linoleic acid improves assembly of the CII subunit and CIII2/CIV complex of the mitochondrial oxidative phosphorylation system in heart failure. Cell Commun Signal. 2019;17(1):128.
- Gonzalvez F, et al. Barth syndrome: cellular compensation of mitochondrial dysfunction and apoptosis inhibition due to changes in cardiolipin remodeling linked to tafazzin (TAZ) gene mutation. Biochim Biophys Acta. 2013;1832(8):1194–1206.
- Xu Y, et al. Remodeling of cardiolipin by phospholipid transacylation. J Biol Chem. 2003;278(51):51380–51385.
- Dudek J, Maack C. Barth syndrome cardiomyopathy. Cardiovasc Res. 2017;113(4):399–410.
- Sack MN, et al. Fatty acid oxidation enzyme gene expression is downregulated in the failing heart. Circulation. 1996;94(11):2837–2842.
- Karbowska J, et al. Peroxisome proliferator-activated receptor alpha is downregulated in the failing human heart. Cell Mol Biol Lett. 2003;8(1):49–53.View this article via: PubMed Google Scholar
- Lionetti V, et al. Carnitine palmitoyl transferase-I inhibition prevents ventricular remodeling and delays decompensation in pacing-induced heart failure. Cardiovasc Res. 2005;66(3):454–461.
- Turcani M, Rupp H. Etomoxir improves left ventricular performance of pressure-overloaded rat heart. Circulation. 1997;96(10):3681–3686.
- Lopaschuk GD, et al. Myocardial fatty acid metabolism in health and disease. Physiol Rev. 2010;90(1):207–258.
- Taegtmeyer H, et al. Assessing cardiac metabolism: a scientific statement from the American Heart Association. Circ Res. 2016;118(10):1659–1701.
- Mizuno Y, et al. The diabetic heart utilizes ketone bodies as an energy source. Metabolism. 2017;77:65–72.
- Osorio JC, et al. Impaired myocardial fatty acid oxidation and reduced protein expression of retinoid X receptor-alpha in pacing-induced heart failure. Circulation. 2002;106(5):606–612.
- Hermann H-P, et al. Haemodynamic effects of intracoronary pyruvate in patients with congestive heart failure: an open study. Lancet. 1999;353(9161):1321–1323.
- Nikolaidis LA, et al. Recombinant glucagon-like peptide-1 increases myocardial glucose uptake and improves left ventricular performance in conscious dogs with pacing-induced dilated cardiomyopathy. Circulation. 2004;110(8):955–961.
- Liao R, et al. Cardiac-specific overexpression of GLUT1 prevents the development of heart failure attributable to pressure overload in mice. Circulation. 2002;106(16):2125–2131.
- Morrow DA, Givertz MM. Modulation of myocardial energetics: emerging evidence for a therapeutic target in cardiovascular disease. Circulation. 2005;112(21):3218–3221.
- Faadiel Essop M, Opie LH. Metabolic therapy for heart failure. Eur Heart J. 2004;25(20):1765–1768.
- Lopaschuk GD, et al. Etomoxir, a carnitine palmitoyltransferase I inhibitor, protects hearts from fatty acid-induced ischemic injury independent of changes in long chain acylcarnitine. Circ Res. 1988;63(6):1036–1043.
- Rehmani T, et al. Cardiac-specific cre induces age-dependent dilated cardiomyopathy (DCM) in mice. Molecules. 2019;24(6):1189.
- Pugach EK, et al. Prolonged Cre expression driven by the α-myosin heavy chain promoter can be cardiotoxic. J Mol Cell Cardiol. 2015;86:54–61.
- Lygate CA. The pitfalls of in vivo cardiac physiology in genetically modified mice - lessons learnt the hard way in the creatine kinase system. Front Physiol. 2021;12:685064.
- Goldberg Ira J, et al. Lipid metabolism and toxicity in the heart. Cell Metab. 2012;15(6):805–812.
- Kienesberger PC, et al. Myocardial triacylglycerol metabolism. J Mol Cell Cardiol. 2013;55:101–110.
- Lee HJ, et al. Selective remodeling of cardiolipin fatty acids in the aged rat heart. Lipids Health Dis. 2006;5:2.
- Zhu S, et al. Cardiolipin remodeling defects impair mitochondrial architecture and function in a murine model of barth syndrome cardiomyopathy. Circ Heart Fail. 2021;14(6):e008289.
- Yazawa E, et al. A murine model of Barth syndrome recapitulates human cardiac and skeletal muscle phenotypes. Dis Model Mech. 2025;18(5):dmm052077.
- Dudek J, et al. Cardiac-specific succinate dehydrogenase deficiency in Barth syndrome. EMBO Mol Med. 2016;8(2):139–154.
- Powers C, et al. Diminished exercise capacity and mitochondrial bc1 complex deficiency in tafazzin-knockdown mice. Front Physiol. 2013;4:74.
- Chandler MP, et al. Moderate severity heart failure does not involve a downregulation of myocardial fatty acid oxidation. Am J Physiol Heart Circ Physiol. 2004;287(4):1538–1543.
- Cahill GF, JrFuel metabolism in starvation. Annu Rev Nutr. 2006;26:1–22.
- Janardhan A, et al. Altered systemic ketone body metabolism in advanced heart failure. Tex Heart Inst J. 2011;38(5):533–538.View this article via: PubMed Google Scholar
- Ferrannini E, et al. CV protection in the EMPA-REG OUTCOME trial: a “thrifty substrate” hypothesis. Diabetes Care. 2016;39(7):1108–1114.
- Elnwasany A, et al. Reciprocal regulation of cardiac β-oxidation and pyruvate dehydrogenase by insulin. J Biol Chem. 2024;300(7):107412.
- Sullivan EM, et al. Docosahexaenoic acid lowers cardiac mitochondrial enzyme activity by replacing linoleic acid in the phospholipidome. J Biol Chem. 2018;293(2):466–483.
- Ting HC, et al. Double bonds of unsaturated fatty acids differentially regulate mitochondrial cardiolipin remodeling. Lipids Health Dis. 2019;18(1):53.
- Claypool SM, Koehler CM. The complexity of cardiolipin in health and disease. Trends Biochem Sci. 2012;37(1):32–41.
- Liang G, et al. Diminished hepatic response to fasting/refeeding and liver X receptor agonists in mice with selective deficiency of sterol regulatory element-binding protein-1c. J Biol Chem. 2002;277(11):9520–9528.
- Schiattarella GG, et al. Nitrosative stress drives heart failure with preserved ejection fraction. Nature. 2019;568(7752):351–356.
- Moon YA, et al. Deletion of ELOVL5 leads to fatty liver through activation of SREBP-1c in mice. J Lipid Res. 2009;50(3):412–423.
- Millington DS, et al. Tandem mass spectrometry: a new method for acylcarnitine profiling with potential for neonatal screening for inborn errors of metabolism. J Inherit Metab Dis. 1990;13(3):321–324.
- Giesbertz P, et al. An LC-MS/MS method to quantify acylcarnitine species including isomeric and odd-numbered forms in plasma and tissues. J Lipid Res. 2015;56(10):2029–2039.
-
Version history
- Version 1 (May 1, 2026): Electronic publication



Copyright © 2026 American Society for Clinical Investigation
ISSN: 0021-9738 (print), 1558-8238 (online)








