Review
Abstract
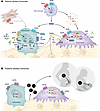
Breakthroughs in rare genetic disease research elucidate the relationships among cytosolic DNA sensing, genome instability, and autoimmune disease phenotypes. Cytosolic self-DNA is a potent trigger of innate immunity, activating the DNA sensor cyclic GMP-AMP synthase (cGAS) and its downstream effector stimulator of interferon genes (STING). This pathway is negatively regulated by the DNA-degrading enzyme three-prime repair exonuclease 1 (TREX1); loss-of-function TREX1 variants lead to accumulation of cytosolic DNA, resulting in STING-mediated autoinflammation. Similarly, STING gain-of-function mutations cause STING-associated vasculopathy with onset in infancy, another disease characterized by multi-organ damage, disability, and premature death. The TREX1-cGAS-STING pathway has also been implicated in regulation of genome stability. Indeed, DNA damage lies at the heart of a separate TREX1-mediated disease, known as retinal vasculopathy with cerebral leukoencephalopathy, where the aberrant nuclear activity of mislocalized TREX1 damages genomic DNA, resulting in multi-organ degeneration syndrome with features of autoimmunity. Thus, monogenic autoimmune diseases and DNA damage syndromes sometimes overlap clinically, and the study of these diseases has created pathways for developing first-in-class small molecule therapeutics.
Authors
Debby J. Park, Kate M. Jones, Jessica B. Anderson, Amanda V. Finck, Jonathan J. Miner
Abstract
Identification of the genetic mutations underlying the ultrarare monogenic conditions STING-associated vasculopathy with onset in infancy (SAVI) and coatomer protein complex subunit alpha (COPA) syndrome revealed a role for the stimulator of interferon genes (STING) immune pathway in the pathogenesis of interstitial lung disease (ILD) in these conditions. STING-focused therapeutics could be a potential avenue for the treatment of SAVI and COPA syndrome in the future, yet the relevance of STING to more common types of ILD is not clear. Here, we provide an overview of SAVI and COPA syndrome, the nature of ILD in these conditions, and current evidence regarding STING activity in their pathogenesis. We discuss data from studies of a variety of other ILDs and model systems and explore the potential role for STING in more common forms of ILD.
Authors
Prasad Palani Velu, Gaofeng Zhu, Karen J. Mackenzie
Abstract
The cyclic GMP-AMP synthase–stimulator of interferon genes (cGAS-STING) pathway is a central mediator of cytosolic DNA–induced innate immune responses, driving the production of type I IFNs and pro-inflammatory cytokines. Beyond its canonical role in cytosolic DNA sensing, increasing attention has been directed toward the noncanonical functions of cGAS and STING, particularly within the nucleus. Recent studies implicate dysregulated cGAS-STING signaling in neurodegenerative diseases and brain aging, with a prominent contribution to glial activation–associated neuroinflammation, a hallmark of many neurological disorders. In this Review, we first summarize the molecular mechanisms underlying the canonical cGAS-STING pathway in DNA sensing and innate immune activation. We then discuss emerging noncanonical roles of cGAS in chromatin organization and RNA metabolism, drawing on insights from evolutionary conservation and protein interactome analyses. Finally, we outline the involvement of cGAS-STING signaling in diverse aspects of brain function, including glial state regulation, neuronal homeostasis, blood-brain barrier integrity, and peripheral immune surveillance, highlighting their contributions to neuroinflammation and neuropathology. We also summarize current pharmacological inhibitors targeting cGAS and STING and discuss their therapeutic potential for modulating cGAS-STING signaling to manage brain disorders.
Authors
Weixi Feng, Abulimiti Aikedan, Subhash C. Sinha, Li Gan
Abstract
Recent advances in cerebrovascular genomics, single-cell biology, pharmacology, and gene editing technology are transforming our understanding of brain arteriovenous malformations (bAVMs) — a leading cause of pediatric hemorrhagic stroke. Once considered static anatomical defects, bAVMs are now recognized as dynamic, genetically driven lesions resulting from somatic mutations in KRAS, BRAF, and pathways involved in arteriovenous specification, angiogenesis, and vascular remodeling. By integrating human genetics, animal models, and endovascular innovations, researchers have uncovered convergent mechanisms that link endothelial Ras/MAPK hyperactivation to abnormal vessel growth and higher rupture risk. These insights provide a foundation for precision medicine approaches that combine molecular diagnostics — such as liquid or endoluminal biopsies — with mutation-specific pharmacotherapies and emerging CRISPR-based gene editing strategies. We suggest that genotype-guided interventions, tailored by spatial and developmental cerebrovascular context, could ultimately reclassify bAVMs from surgically incurable malformations to treatable molecular conditions.
Authors
Andrew T. Hale, Adam J. Kundishora, Pazhanichamy Kalailingam, Tanyeri Barak, Phan Q. Duy, Christopher M. Ramundo, Baojian Fan, Qiang Li, Priscilla K. Brastianos, Ganesh M. Shankar, Seth L. Alper, Benjamin P. Kleinstiver, Patricia L. Musolino, Kristopher T. Kahle
Abstract
Type 1 diabetes mellitus (T1D) has been recognized as a chronic autoimmune disease for five decades, but therapy has relied on the exogenous replacement of insulin, which is an imperfect substitute for normal β cell function. In recent years, there has been progress in the development of new therapeutics that target the primary causes of the disease: failed immunologic tolerance and β cell killing. One agent, teplizumab, was shown to attenuate loss of β cell function that occurs over time and delay progression to clinical disease in individuals at risk, leading to its regulatory approval in 2022. Other immunologic agents show promise in modulating the immunologic imbalance. Moreover, a role for β cells in T1D pathogenesis has been identified and may be targeted. Now that the first disease-modifying therapeutic agent is available, future studies may involve combinations of agents to extend immunologic tolerance and protect and restore β cells so that lasting metabolic remission can be achieved.
Authors
Kevan C. Herold, Carmella Evans-Molina
Abstract
Anemia affects one-third of the population globally and is marked by impaired erythropoiesis that results in substantial mortality and morbidity. Over the past few decades, our understanding of the molecular mechanisms underlying anemia has progressed but translating that knowledge into effective targeted therapeutics remains challenging. Preclinical and clinical studies substantiate the efficacy of modulating erythropoietin-driven signaling pathways to stimulate erythropoiesis. Additional approaches include strategies to maintain iron homeostasis and control iron metabolism, using small molecules and oral supplements. New frontiers in molecular regulation of anemia include perturbation of regulatory genes and spliceosome proteins in erythroid cells, as well as mutation-specific therapeutic approaches. Finally, new evidence supporting the importance of neuronal signaling and mitochondrial dynamics in shaping erythropoiesis is pointing toward novel interventions. Here, we discuss the molecular and genetic factors underlying defective erythropoiesis and highlight current and emerging therapies, including molecular targets to overcome drug resistance.
Authors
Nilesh Rai, Omar Abdel-Wahab, Lingbo Zhang
Abstract
Bone is a highly dynamic and purposefully organized structure that remodels constantly throughout adult life. Disordered bone remodeling, in which resorption of old bone by osteoclasts exceeds new bone formation by osteoblasts, results in bone loss, which, in turn, is associated with debilitating conditions, including osteoporosis and metastatic bone disease. The past decade has revealed vital new insights into the role of the central nervous system in skeletal regulation. These studies have led to a better understanding of physiologic circuitry, enabled us to revisit disease pathophysiology, and in doing so, prompted the creation of candidate therapeutics. The central neural control of bone is exerted through two arms — an amplitude-modulated (AM) neurohormonal arm that relies on changes in circulating levels of anterior and posterior pituitary hormones, which act on bone directly, and a frequency-modulated (FM) arm that arises from changes in the firing frequency of sympathetic, parasympathetic, and sensory nerves that innervate bone. Here, we review the medical consequences arising from the dysfunction of the AM and FM arms, as well as studies that have unmasked promising therapeutic targets.
Authors
Mone Zaidi, Se-Min Kim, Vitaly Ryu, Daria Lizneva, Terry F. Davies, Clifford J. Rosen, Tony Yuen, Andrea Giustina
Abstract
Although combination antiretroviral therapy (ART) has dramatically reduced the incidence of severe HIV-associated neurological disease, the central nervous system (CNS) remains a viral sanctuary in which inflammation and brain injury persist despite systemic viral suppression. Here, we synthesize evidence that ongoing HIV-associated brain injury is sustained not primarily by unchecked viral replication but by persistent viral transcription from defective proviruses, immune-mediated synaptic dysfunction driven by bystander activation, and long-lived microglial reprogramming shaped by epigenetic “training.” We highlight how emerging single-cell multiomics and “liquid biopsy” approaches are redefining our understanding of the CNS reservoir at high resolution. We further discuss the growing emphasis on biologically anchored, molecularly defined disease subtypes as a means to disentangle HIV-specific pathology from the confounding overlap of aging and multimorbidity, which have increased in the ART era. Finally, we underscore the necessity of human-centered translational studies to validate preclinical findings, outlining how these molecular insights pave the way for precision therapeutics and CNS-targeted cure strategies.
Authors
Paraskevas Filippidis, Shelli F. Farhadian
Abstract
Neurodegenerative diseases arise from interactions among pathogenic proteins, immune responses, and diverse environmental or age-related stressors that disrupt CNS homeostasis. CNS resident microglia detect self-derived danger signals through pattern recognition receptors, and their activation can promote clearance of aberrant proteins, including amyloid-β, tau, α-synuclein, and TAR DNA-binding protein 43. However, microglial activation may also drive maladaptive states that amplify neuroinflammation. Microglial transitions are further shaped by receptor-mediated signaling and antigen presentation pathways that integrate environmental cues with functional responses. Adaptive immune cells contribute additional layers of regulation, with CD8+ and CD4+ T cells exerting neuroprotective or neurotoxic effects depending on disease context, activation state, and antigen specificity. The identification of granzyme K–expressing CD8+ T cells in several neurodegenerative conditions highlights the growing recognition that distinct T cell subsets may have specialized roles in disease. Aging, repetitive head injury, and viral infection further alter microglial phenotypes, weaken barrier integrity, promote T cell recruitment, and prime the CNS for chronic inflammation. In this review, we synthesize current knowledge of innate and adaptive immune mechanisms in neurodegeneration, examine how external factors influence these responses, and consider how these insights may guide future therapeutic strategies.
Authors
Yvonne L. Latour, Dorian B. McGavern
Abstract
Lysosomes function as metabolic control centers that integrate degradation, nutrient sensing, and stress signaling. In neurons, which must maintain proteostasis and energetic balance throughout life, lysosomal homeostasis determines cellular resilience. Emerging evidence identifies lysosomal injury and defective repair as common denominators across neurodegenerative diseases. Damage to the lysosomal membrane caused by oxidative stress, lipid imbalance, or genetic mutations triggers a hierarchical quality control cascade. Early lesions recruit the endosomal sorting complex required for transport (ESCRT) machinery for mechanical resealing, while larger ruptures activate lipid-centered recovery modules. When repair fails, lysophagy eliminates irreparable organelles and a TFEB-dependent transcriptional program regenerates the lysosomal pool. These tightly coupled responses safeguard neurons from catastrophic proteostatic collapse. Their impairment, through mutations in lysosomal proteins, or through aging, produces the lysosomal fragility that underlies Alzheimer disease, Parkinson disease, amyotrophic lateral sclerosis/frontotemporal dementia, and Huntington disease. Crosstalk between lysosomes, mitochondria, and ER integrates local damage with systemic metabolic adaptation, while dysregulated lysosomal exocytosis and inflammation propagate pathology. Understanding how ESCRT complexes, lipid transport, and transcriptional renewal cooperate to preserve lysosomal integrity reveals unifying principles of neurodegeneration and defines molecular targets for intervention. Restoring lysosomal repair and renewal offers a rational path toward preventing neuronal loss.
Authors
Stefano De Tito, Sharon A. Tooze
No posts were found with this tag.