Advertisement
Review
Open Access |  10.1172/JCI204831
10.1172/JCI204831
Smoldering in the sanctuary: HIV-associated brain injury in the ART era
Paraskevas Filippidis1,2 and Shelli F. Farhadian3,4
1Department of Pathology, Yale School of Medicine, New Haven, Connecticut, USA.
2Infectious Diseases Service, Department of Medicine, Lausanne University Hospital and University of Lausanne, Lausanne, Switzerland,
3Department of Internal Medicine, Section of Infectious Diseases, and
4 Department of Neurology, Yale School of Medicine, New Haven, Connecticut, USA.
Address correspondence to: Shelli F. Farhadian, Yale School of Medicine, 300 George Street, New Haven, Connecticut 06520, USA. Phone: 203.785.2647; Email: shelli.farhadian@yale.edu.
Find articles by Filippidis, P. in: PubMed | Google Scholar
1Department of Pathology, Yale School of Medicine, New Haven, Connecticut, USA.
2Infectious Diseases Service, Department of Medicine, Lausanne University Hospital and University of Lausanne, Lausanne, Switzerland,
3Department of Internal Medicine, Section of Infectious Diseases, and
4 Department of Neurology, Yale School of Medicine, New Haven, Connecticut, USA.
Address correspondence to: Shelli F. Farhadian, Yale School of Medicine, 300 George Street, New Haven, Connecticut 06520, USA. Phone: 203.785.2647; Email: shelli.farhadian@yale.edu.
Find articles by
Farhadian, S.
in:
PubMed
|
Google Scholar
|

Published May 1, 2026 - More info
J Clin Invest. 2026;136(9):e204831. https://doi.org/10.1172/JCI204831.
© 2026 Filippidis, et al. This work is licensed under the Creative Commons Attribution 4.0 International License. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
-
Abstract
Although combination antiretroviral therapy (ART) has dramatically reduced the incidence of severe HIV-associated neurological disease, the central nervous system (CNS) remains a viral sanctuary in which inflammation and brain injury persist despite systemic viral suppression. Here, we synthesize evidence that ongoing HIV-associated brain injury is sustained not primarily by unchecked viral replication but by persistent viral transcription from defective proviruses, immune-mediated synaptic dysfunction driven by bystander activation, and long-lived microglial reprogramming shaped by epigenetic “training.” We highlight how emerging single-cell multiomics and “liquid biopsy” approaches are redefining our understanding of the CNS reservoir at high resolution. We further discuss the growing emphasis on biologically anchored, molecularly defined disease subtypes as a means to disentangle HIV-specific pathology from the confounding overlap of aging and multimorbidity, which have increased in the ART era. Finally, we underscore the necessity of human-centered translational studies to validate preclinical findings, outlining how these molecular insights pave the way for precision therapeutics and CNS-targeted cure strategies.
-
Introduction
Neurologic complications were among the earliest and most devastating manifestations of HIV infection, recognized within the first years of the epidemic as sentinel features of advanced disease (1, 2). In the era before antiretroviral therapy (ART), the burden of central nervous system (CNS) involvement was striking. HIV-associated dementia developed in approximately 15%–20% of individuals with advanced AIDS, and milder neurocognitive syndromes were observed in up to 30%–50% of patients with uncontrolled viremia (3, 4). Neurologic decline often paralleled immunologic collapse, and the presence of dementia portended shortened survival. These observations firmly established the brain as a major target of HIV pathogenesis and positioned neuroAIDS as a defining complication of untreated infection (5, 6).
The advent of combination ART dramatically altered this landscape. Effective systemic viral suppression has led to a marked decline in the incidence of HIV-associated dementia and other severe opportunistic neurologic syndromes. In contemporary cohorts, frank HIV dementia is now rare in high-resource settings where early diagnosis and durable viral suppression are common (3). However, the elimination of severe dementia has not equated to restoration of normal brain health. Depending on cohort characteristics and diagnostic criteria, 10%–60% of people with HIV (PWH) continue to meet criteria for some form of neurocognitive impairment (NCI), though the clinical relevance and biological specificity of these classifications remain debated (7–10). Even among virally suppressed individuals who are neurologically asymptomatic, structural neuroimaging abnormalities, cerebrospinal fluid (CSF) immune activation, and molecular evidence of CNS injury persist (11–14). These observations underscore a central paradox of the ART era: while systemic replication is effectively controlled, the CNS remains biologically perturbed.
In parallel, the demographic transformation of the HIV epidemic — with increasing life expectancy — has introduced the intersecting influences of aging, vascular disease, substance use, polypharmacy, and social determinants of health, all of which independently affect brain integrity (15–21). As a result, contemporary HIV-associated CNS disease reflects not only residual viral effects but also the convergence of viral legacy, immune dysregulation, and age-related comorbid processes.
In this Review, we examine HIV neuropathogenesis across the treatment continuum, contrasting the catastrophic inflammatory encephalitis of the pre-ART era with the subtler yet biologically active injury that characterizes treated infection. Rapid advances in single-cell multiomics, spatial transcriptomics, ultrasensitive reservoir assays, and multimodal neuroimaging are transforming our ability to interrogate the CNS reservoir and its downstream consequences in living individuals. Here we synthesize current understanding of how persistent viral transcription from intact and defective proviruses, immune-mediated synaptic dysfunction, and epigenetically reprogrammed microglia sustain CNS injury despite ART. Finally, we discuss evolving conceptual frameworks — including HIV-associated brain injury biotypes — and emerging translational approaches that aim to move the field from descriptive epidemiology toward mechanism-based precision therapeutics.
-
HIV’s impact on the CNS before treatment
CNS viral entry and infection persistence. HIV enters the CNS within days to weeks of systemic infection (22). The predominant mechanism of HIV neuroinvasion likely involves the transendothelial migration of infected immune cells, a process augmented by HIV gp120’s deleterious effects on tight junctions of the blood-brain barrier (23). Early studies, using indirect methods such as in vitro assays and peripheral blood immunophenotyping, suggested that monocytes — particularly CD14+CD16+ subsets — were the primary immune cell type carrying HIV to the CNS during early infection (24–26). Likewise, in autopsy studies of both simian immunodeficiency virus–infected primates and individuals with HIV, replicating virus in the CNS is detected primarily in perivascular (monocyte-derived) macrophages and microglia (27, 28), further implicating myeloid cells in establishing and sustaining HIV infection in the CNS.
While monocyte/macrophage trafficking represents the canonical route of HIV neuroinvasion, accumulating evidence indicates that infected CD4+ T cells further contribute to the establishment and sustainment of CNS infection (as reviewed in ref. 29). Phylogenetic analyses of paired CSF and plasma viral sequences demonstrate that early CNS viral populations require cells with high CD4 density for cell entry (a density found on T cells but not myeloid lineage cells) and that these sequences are genetically similar to T cell–tropic strains (30). Viral strains capable of infecting T cells but not monocytes precede the emergence of strains capable of infecting cells with low CD4 density, such as macrophages, later in infection, and support a model in which infected T cells establish the first wave of CNS infection (31). Macrophages may also become infected through the capture and engulfment of HIV-infected CD4+ T cells (32). Evidence from human postmortem and CSF studies further shows that HIV DNA is detectable in CNS-infiltrating T cells before treatment, underscoring a critical role of T cells in founding the CNS reservoir (33, 34).
Once HIV is established in the CNS, it is sustained through localized replication within the CNS, in addition to ongoing trafficking of infected cell–associated and cell-free virus from the periphery. Phylogenetic analyses performed on specimens obtained from viremic individuals demonstrate that HIV sequences isolated from CSF often diverge from contemporaneous plasma sequences, providing evidence of localized viral replication and adaptation within the CNS (31).
Collectively, these findings indicate that infected T cells play a key role in the earliest stages of HIV neuroinvasion, seeding infection in the brain that is later maintained and amplified by macrophages and microglia. This knowledge has informed the basis for current studies that focus on macrophages and microglia as the primary source of residual viral RNA and protein production in PWH who are on suppressive ART.
Neuropathological impact of HIV in the brain. Autopsy studies from the pre-ART era provide the clearest insights into how sustained, uncontrolled HIV replication damages the CNS. Brain tissue from many individuals who died with untreated HIV infection exhibit a characteristic constellation of pathological findings collectively termed HIV encephalitis (HIVE). HIVE is defined by microglial nodules containing HIV antigens, astroglial activation, myelin loss, and brain atrophy (1, 35). These lesions are most prominent in deep gray matter and subcortical white matter, and their presence strongly associates with clinical HIV-associated dementia before death (2, 36). Although neurons are rarely infected, autopsy investigations consistently demonstrate loss of synapses and dendritic architecture, as well as axonal injury, supporting the model that neurodegeneration arises largely from inflammatory and virally toxic cascades rather than direct neuronal infection (37). Beyond HIVE, several forms of white-matter pathology were frequently observed in pre-ART specimens, including vacuolar encephalopathy, consisting of intramyelinic vacuoles with relatively intact axons and diffuse leukoencephalopathy (38).
Importantly, many individuals with severe cognitive impairment had no or only minimal discrete lesions visible on routine brain histology (5, 6). When compared with the brains of PWH with NCI and HIVE, these brains show different transcriptional and protein profiles, implying different biological mechanisms underlying similar clinical phenotypes (39, 40).
Phylogenetic studies of virus within distinct brain regions further shows that HIV replication varies markedly across brain regions and evolves independently within the CNS, reinforcing the concept of compartmentalization in natural history infection (41). Thus, early autopsy studies established the CNS as both a key target and reservoir of HIV, with inflammation-driven neuronal and synaptic damage underlying the high burden of cognitive impairment prior to effective therapy.
-
HIV in the brain after ART: host immunity
The clinical expression of HIV-associated CNS disease has changed dramatically with the advent of potent ART. Although severe HIV-associated CNS diseases such as HIV dementia are now rare (42), a substantial subset of PWH continue to exhibit persistent CNS immune activation, microglial dysregulation, and cognitive or neuropsychiatric symptoms despite long-term viral suppression. Modern work — including structural neuroimaging, CSF biomarker profiling, and single-cell immunologic studies — has shifted the focus from uncontrolled viral replication to the mechanisms by which HIV alters CNS biology despite ART.
CSF biomarkers of residual neuroinflammation and injury. Despite durable viral suppression with ART, persistent CNS immune activation remains a hallmark of HIV neuropathogenesis and is consistently linked to NCI. Much of the evidence for ongoing neuroimmune dysfunction derives from CSF studies. It is important to note that CSF is not a direct surrogate of brain parenchyma, but rather acts as a dynamic interface enriched for immune surveillance, trafficking cells, and soluble mediators that integrate signals from multiple CNS compartments. These CSF studies of PWH on ART demonstrate sustained elevations in monocyte/macrophage activation markers (e.g., neopterin, soluble CD14, soluble CD163) and inflammatory cytokines and chemokines (e.g., IFN-γ, IL-1α, CCL2) (11), often paired with markers of glial activation, neuronal injury, and neurodegeneration such as neurofilament light chain, YKL-40, TREM2, and glial fibrillary acidic protein (12, 13). Systematic reviews and targeted studies identify a core set of CSF analytes — including CCL2/monocyte chemoattractant protein-1, IL-8/CXCL8, and CXCL10/IP-10 — associated with NCI, though effect sizes and directions vary, reflecting biological heterogeneity and limitations of clinical phenotyping (11, 43). High-throughput proteomic approaches have further reinforced this framework, revealing broad signatures of residual CNS immune activation and axonal injury that persist despite ART (44). Ultrasensitive platforms such as single-molecule array assays now enable detection of low-abundance CNS injury biomarkers in blood and CSF and may play an important role in future longitudinal and interventional studies. However, soluble biomarkers alone provide an indirect and sometimes inconsistent view of disease mechanisms, underscoring the need to integrate these findings with insights from cellular analyses to more precisely define the immune drivers of ongoing CNS injury in treated HIV.
Persistent microglial activation despite ART. Within the CNS, two principal macrophage populations drive HIV pathogenesis: yolk-sac–derived parenchymal microglia, which self-renew locally, and border-associated macrophages (perivascular, meningeal, and choroid plexus), which are periodically replenished by circulating monocytes (45–47). Together, these CNS myeloid populations serve as central cellular targets and long-lived reservoirs for HIV.
Even under suppressive ART, these cells sustain neuropathogenesis through both direct infection and indirect “bystander” activation. While infected microglia (estimated to comprise less than 0.5% of the total population) (48) specifically upregulate S100 family genes (which modulate inflammatory responses in the CNS) (49, 50), and various chemokines and IFN-related pathways, uninfected bystander cells also adopt inflammatory phenotypes, responding to local inflammatory cues, such as viral proteins, and leading to synaptodendritic injury (48, 50) (Figure 1). This is further corroborated by studies using translocator protein (TSPO) PET imaging. TSPO is expressed on activated glia; increased TSPO radiotracer binding is consistently found in PWH on suppressive ART compared with controls, even in asymptomatic individuals (51, 52). It should be noted, though, that the cellular specificity of TSPO in human studies is limited, as TSPO is also expressed by astrocytes and endothelial cells. This underscores the need to interpret TSPO signal as a composite marker of neuroinflammation and highlights the importance of developing next-generation tracers capable of distinguishing microglial from astroglial activation in vivo.
 Figure 1
Figure 1The “smoldering secretome”: mechanisms of bystander injury during viral suppression. Even in the absence of productive viral replication, HIV reservoirs and persistent immune activation in the CNS drive ongoing neurotoxicity. The “smoldering” reservoir microglia (left) harbors defective or silenced proviruses that retain the capacity to transcribe viral RNA and produce viral proteins (such as Tat, Nef, or gp120) without assembling infectious virions. These viral products, along with cell-associated RNA (CA-RNA), are released into the extracellular space as soluble molecules or packaged into EVs. Adaptive immune cells also contribute to this compartmentalized inflammation. Activated CD4+ T cells, which may also harbor HIV DNA, interact with microglia and may release both pro-inflammatory cytokines and viral transcripts or proteins. Simultaneously, activated CD8+ T cells may play a role both in viral clearance and in fueling inflammation by secreting soluble inflammatory factors, such as IFN-γ. This “viral secretome” traffics to uninfected bystander cells, including microglia and neurons. Uptake of these toxic EVs triggers inflammasome activation and cytokine release in bystander microglia and induces synaptic damage, sustaining neuroinflammation and brain injury despite ART.
Recent single-nucleus RNA-seq (snRNA-seq) and ATAC-seq studies demonstrate that chronic HIV drives 3D chromatin reconfiguration and promotes pro-inflammatory, neurodegeneration-related, and senescent gene programs even in the absence of overt viral replication (48, 53). A key driver of this primed state is the NLRP3 inflammasome. Viral proteins Tat, gp120, and Vpr prime and activate the inflammasome, promoting caspase-1 cleavage and release of IL-1β and IL-18 (54–58). These processes recruit additional immune cells and propagate inflammatory signaling to bystander neurons and glia, further amplifying neurodegeneration. Mechanistically, this persistence of abnormal microglial proliferation and activation may be driven by trained immunity — a process of long-term functional reprogramming of innate immune cells via epigenetic remodeling (59); however, further studies are required to understand the role of epigenetic alterations in HIV-associated microglial activation.
Beyond bona fide resident brain microglia, monocyte-derived microglia-like cells are present in the adult human brain and in CSF and may further contribute to HIV neuropathogenesis (60). Recent CSF profiling studies have identified CD204 as a marker to identify and isolate these cells from the CSF of PWH (14, 61). Thus, CD204-expressing cells in CSF may provide a window into microglial activation states in vivo and represent a promising candidate biomarker for HIV-associated brain injury (14, 61). However, further immunophenotyping is required to define their precise role in HIV pathology.
Cell-mediated immunity. HIV is fundamentally T cell tropic, but CNS impairment extends beyond the direct effects of CD4+ T cell depletion. Even in the setting of durable virologic suppression, trafficking, activation, and functional skewing of lymphocytes within the CNS sustain compartmentalized neuroinflammation (14, 62, 63). Critically, because brain tissue is rarely sampled, our understanding of these dynamics relies heavily on the study of CSF-derived cells.
In the CNS, CD8+ T cells play a dual role. While HIV-specific CD8+ T cells — including highly activated and MHC class I–restricted CD4dimCD8bright subsets — limit viral replication and correlate with reduced inflammation (64), generalized CD8+ T cell activation drives pathogenesis. Longitudinal and single-cell RNA-seq (scRNA-seq) analyses show that activated CD8+ T cell phenotypes persist in CSF despite treatment (14, 63, 65). This chronic activation, alongside CD4/CD8 imbalance and increased IFN-γ production, correlates with cognitive impairment and white-matter abnormalities on MRI (62, 66). A similar process is observed in peripheral blood, where T cell senescence and exhaustion associate with both cognitive impairment and brain atrophy (67, 68).
Humoral immunity. B cells are increasingly recognized as active participants in shaping CNS inflammation in ART-treated PWH. In the periphery, deficits in resting memory B cells and impaired antigen-specific responses are present despite ART (69, 70). Likewise in the CSF, despite systemic viral suppression, the B cell chemoattractant CXCL13 is frequently elevated and associates with inflammatory biomarkers and cognitive impairment (71). Similarly, intrathecal IgG synthesis is elevated and correlates with both blood-CSF barrier dysfunction and NCI (72, 73), suggesting persistent intrathecal B cell activation and antibody production as potential drivers of CNS inflammation, independent of plasma HIV replication.
High-resolution antibody profiling further shows that CSF antibodies are distinct from those in the periphery — enriched for HIV-specific reactivity but functionally impaired — and are resistant to ART-induced changes (74). Not all dysregulated antibody responses are HIV specific, though: intrathecal anti-EBV IgG and autoantibodies (e.g., anti-CD4) in the CSF correlate with markers of neuroinflammation, neuronal injury, and clinical symptoms (75–77). Collectively, these findings support a model in which persistent B cell activation, intrathecal antibody production, and immune complexes may interact with activated T cells and microglia — via antigen presentation, Fc receptor engagement, and chemokine signaling — to sustain compartmentalized neuroinflammation in the ART era. Further studies characterizing B cell phenotypes and functions beyond antibody production remain essential to refine this model and define new therapeutic targets.
Nonimmune mechanisms of HIV-associated brain injury. Immunologic host factors represent a major focus in HIV neuropathogenesis; however, these processes occur within a broader landscape of interacting nonimmune mechanisms that also contribute to brain vulnerability. Emerging evidence highlights roles for mTOR pathway dysregulation, which affects autophagy, apoptosis, and neuroimmune interactions (78, 79); Alzheimer’s disease–like pathogenic processes, including alterations in amyloid and tau pathways that may be accentuated in aging PWH (80–82); mitochondrial dysfunction, impairing neuronal bioenergetics and promoting oxidative stress (83–85); and gut microbiome perturbations, which fuel systemic immune activation and altered neurotransmitter signaling (86–88). These mechanisms provide complementary frameworks for understanding the multifactorial nature of HIV-associated brain injury; however, a detailed discussion of them is beyond the scope of the present Review.
-
HIV in the brain after ART: viral products
In the ART era, HIV neuropathogenesis has shifted from acute viral cytopathicity to reservoir-driven neuroimmune dysregulation. Persistent HIV proviral DNA reservoirs within the CNS sustain low-level expression of viral transcripts and proteins despite ART. These residual viral products exert direct neurotoxic effects and chronically engage innate and adaptive immune pathways, resulting in bystander neuronal injury, synaptic dysfunction, and ultimately cognitive impairment (89–91). Indeed, HIV-specific CD8+ T cells remain in the CNS even after years of durable ART (64, 92), suggesting viral protein expression even in the presence of ART and clinically suppressed viral load.
Proviruses in the CNS during ART. The characterization of the HIV-1 reservoir within the CNS has transitioned from simple DNA quantification to sophisticated assays capable of distinguishing between intact and defective proviruses. Current methodologies primarily utilize the Intact Proviral DNA Assay (IPDA) and near-full-length genome sequencing (nFGS) to map this landscape (93, 94). The IPDA employs droplet digital PCR with multiplexed probes targeting the highly conserved packaging signal (Ψ) and envelope (env) regions; proviruses are classified as “intact” when both targets are detected, while those missing one or both are identified as defective (93). For definitive characterization, nFGS is employed as the gold standard, involving limiting dilution of DNA to the single-genome level followed by long-range PCR and deep sequencing to identify lethal defects like internal stop codons and hypermutations (94, 95). Recent studies have successfully applied these techniques to both CSF and postmortem brain tissue, revealing that while the majority of CNS proviruses are defective, a stable population of genome-intact, potentially replication-competent virus persists in microglia and other resident cells despite long-term ART (96, 97). Although overall proviral burden in brain tissue is low compared with lymphoid compartments (98), intact genomes are reproducibly detected across multiple brain regions, including frontal white matter and cortex, and in CSF, where approximately 1.7% of total proviral DNA is intact, irrespective of ART status (96, 99, 100). Critically, CNS proviral burden correlates with cognitive deficit scores, implicating these persistent infected cells in ongoing NCI (101–103).
Most HIV provirus in the brain has been localized to microglia and other myeloid lineage cells (96, 100, 104, 105), with approximately 0.5% of these cells harboring HIV DNA, including inducible replication-competent virus (48, 102, 105, 106). Viral subtype may also influence reservoir burden, with subtype C–infected brains showing lower intact proviral frequencies compared with subtype B–infected brains (107). In addition to resident myeloid cells, a distinct population of CD4dimCD8bright T cells has been shown, in an animal model, to traffic to the brain and harbor replication-competent HIV, suggesting a role for lymphocyte-mediated HIV neuroinvasion and CNS persistence alongside established myeloid reservoirs (108).
The vast majority of cells in the brain are glial cells, predominantly astrocytes. Thus, there has been considerable attention paid to whether these non-CD4-expressing cells are infected by HIV and whether they serve as a nonimmune CNS reservoir. The results are conflicting. Although a seminal RNAscope and DNAscope study of postmortem brains did not detect HIV in any brain astrocytes (102), studies using more sensitive assays have consistently detected integrated HIV DNA in a subset of astrocytes, even among PWH on long-term suppressive ART (97, 109–112). Astrocytes appear to be infected via receptor-mediated endocytosis and by cell-to-cell contact from infected lymphocytes (113). The consequences of the astrocyte reservoir are potentially important: animal studies suggest that astrocytes are capable of producing virions that are trafficked to the periphery via infected CD4+ T cells and may contribute to viral reseeding outside of the CNS (110).
Persistent HIV transcription in the CNS. Although ART blocks productive HIV replication, viral transcription persists in the CNS of virally suppressed PWH. Defective proviruses, long assumed to be silent, dominate brain-resident HIV genomes and retain transcriptional competence, ultimately enabling the production of aberrant or truncated proteins with inflammatory potential (96, 100, 114–117).
In accord with studies of HIV DNA, postmortem studies consistently identify microglia as the dominant source of HIV RNA in the brain (48, 106). These HIV RNA–positive microglia may exhibit a distinct inflammatory phenotype, despite broadly similar chromatin accessibility across microglial subsets; however, larger studies with greater numbers of HIV RNA–positive cells are required to draw more definitive conclusions (48). In frontal cortex tissue from virally suppressed and nonsuppressed individuals, low but detectable HIV transcripts were detected in most ART-treated PWH, in the range of tens to hundreds of copies per 106 cells, with evidence of transcriptional completion in a subset of suppressed individuals (118). Notably, HIV p24+CD68+ myeloid cells were identified in suppressed brains, indicating that viral protein production persists in at least some individuals despite ART. Although a proximal elongation block was observed — more pronounced in virally suppressed participants — these findings suggest that CNS proviruses are transcriptionally restricted rather than fully silenced, reinforcing the concept of a “smoldering” reservoir in the brain capable of sustaining chronic immune activation (118).
In the CSF, HIV RNA is found primarily in central memory CD4+ T cells (14, 119, 120). This discrepancy — with brain studies showing HIV RNA in myeloid cells and CSF showing most HIV transcription in T cells — likely reflects the overall immune cell composition of CSF compared with the brain, with CSF consisting almost entirely (~90%) of T cells (14, 61), compared with very few infiltrating and brain-resident T cells (121). A subset of HIV RNA–containing T cells in the CSF are members of T cell clonal populations present in both blood and CSF (120). The presence of HIV RNA–producing cells sharing TCR sequences across blood and CSF compartments even after decades of ART lends further support to the notion that infected T cells traffic between tissue compartments and that maintenance and expansion of infected T cell clones contribute to the maintenance of the CNS reservoir in PWH on ART.
Beyond cellular transcripts, extracellular vesicles (EVs) in CSF contain multiple classes of HIV RNAs, including readthrough, TAR, long-LTR, Pol, Nef, and Tat–Rev, at higher levels than in blood. These EV-associated transcripts correlate strongly with global and domain-specific cognitive impairment (122).
The synaptoxicity of HIV. EVs containing viral proteins such as Tat, Nef, or gp120 persist in the CNS despite ART (97, 123, 124). In animal models, sustained CNS exposure to gp120 or Tat leads to cognitive impairment (125–128). Henderson et al. detected Tat in the CSF of approximately one-third of ART-treated individuals, packaged within EVs with preserved transactivation activity. Importantly, Tat levels did not correlate with CSF viral load in individuals with transient CSF HIV escape, suggesting that EV-associated Tat may originate from viral reservoirs and be released independently of detectable viral replication (124). Nef, which is also released in EVs from HIV-infected immune and glial cells, has similarly been implicated in CNS injury (129). Although direct infection of microglia can trigger focal release of soluble neurotoxic viral proteins, the widespread synaptic damage observed during ART suppression is likely in large part sustained by EV-mediated transfer of viral proteins to uninfected cells (130–132).
Residual viral proteins drive synaptodendritic injury through multiple, non–mutually exclusive mechanisms. Gp120 and Tat alter NMDA receptor signaling, disrupting calcium homeostasis and promoting neuronal toxicity (133–135). Inflammatory cytokines released from activated bystander microglia, including TNF-α, further exacerbate NMDA receptor dysregulation activity on neurons (136). HIV proteins may also impact the structural stability of dendritic spines by altering host proteins such as the E3 ubiquitin ligase Mdm2 (133, 137). In addition, exposure of uninfected astrocytes to viral proteins impairs glutamate transport, contributing to synaptic loss (138–140).
Together, these findings support a model in which the CNS reservoir functions as a chronic “toxic hub,” where a relatively small population of infected cells drives widespread synaptodegeneration through pathological activation of surrounding uninfected cells. Older ART regimens may also induce synaptic loss (141). However, studies directly assessing synaptic integrity in PWH on suppressive ART remain limited, and much of our understanding of viral protein–mediated synaptic toxicity is derived from animal and in vitro models. Emerging neuroimaging approaches, such as PET imaging with the synaptic vesicle glycoprotein 2A (SV2A) tracer, enable in vivo measurement of synaptic density and may be informative to tracking synaptodendritic injury over the course of HIV and its treatment (142).
-
Clinical challenges and frameworks: HIV, aging, and comorbidities
Evolving concepts: from HAND to HIV-associated brain injury. The historical framework of HIV-associated neurocognitive disorders (HANDs) was developed in the pre-ART era and has limited specificity today. HAND classifications often label 20%–60% of PWH as cognitively impaired, largely because the criteria include asymptomatic neurocognitive impairment, which may lack clinical relevance (3, 4). These prevalence estimates do not align with the clinical experience of providers and likely overestimate clinically meaningful disease in the era of viral suppression.
Given the mismatch between HAND categories and modern biology, there is a shift toward the construct of HIV-associated brain injury, which prioritizes objective evidence of CNS pathology — such as neuroimaging abnormalities, persistent immune activation, and microglial signatures — over neuropsychological thresholds alone (143). This biologically anchored approach aims to categorize PWH into distinct biotypes based on their underlying drivers of injury, such as inflammatory, vascular, or neurodegenerative dominant profiles, acknowledging the multifactorial interplay between HIV legacy effects and non-HIV comorbidities (113–115) (Figure 2).
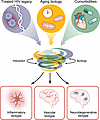 Figure 2
Figure 2Evolving frameworks: the convergence of HIV, aging, and comorbidities into distinct biotypes. Moving beyond the historical HAND classification, modern HIV-associated brain injury is conceptualized as a heterogeneous condition driven by the convergence of three distinct upstream drivers: the legacy of treated HIV (including reservoir persistence, residual inflammation, neuronal injury, and glial activation), the biology of aging (mitochondrial dysfunction, senescence), and the burden of non-AIDS comorbidities (cardiovascular disease, polypharmacy, substance use, coinfections, and social factors). These drivers do not act in isolation but interact synergistically to alter CNS homeostasis. This convergence resolves into phenotypic profiles, or biotypes. Recognizing these complex interactions and biological profiles is essential for identifying risk factors and opportunities for targeted interventions and precision therapeutics.
The aging brain: comorbidities and inflammatory drivers. With increased life expectancy, aging and frailty have emerged as dominant determinants of brain health in PWH. Virally suppressed PWH often exhibit premature or accelerated aging, with higher prevalence and earlier onset of age-associated conditions (15–18, 144–147). Population-level studies using electronic medical record–based diagnoses reveal both a higher prevalence and an earlier age of onset for age-associated dementias in people with HIV, even in the ART era (147, 148). Structural MRI studies support this model, revealing accelerated cortical and white-matter abnormalities in older PWH, particularly among those with high comorbidity burdens (149).
PWH exhibit higher rates of ischemic stroke and cerebral small-vessel disease — including white-matter hyperintensities, lacunar infarcts, and microbleeds — even under viral suppression (150, 151). Mechanistically, chronic inflammation and endothelial dysfunction fuel this damage and delineate a specific neurocognitive profile defined by executive dysfunction and slowed processing speed (152–155).
Stimulants (methamphetamine, cocaine), opioids, and alcohol remain potent amplifiers of host-mediated CNS injury. These substances disrupt blood-brain barrier integrity, enhance microglia driven neuroinflammation, and facilitate the transmigration of HIV-infected monocytes into the CNS (156–158). Chronic coinfections (EBV, CMV, hepatitis C virus) further modulate CNS inflammation (159–162). Sleep disturbance, which is nearly ubiquitous in chronic HIV, may create a feed-forward loop: disrupted sleep impairs glymphatic clearance and alters microglial reactivity, which in turn heightens neuroinflammation and worsens depressive symptoms and cognitive impairment (163–165).
Crucially, the preservation of brain health in PWH is complicated by the cumulative iatrogenic burden of polypharmacy. As individuals age with HIV, the number of prescribed medications increases substantially, often exceeding that of HIV-negative peers, and polypharmacy has been independently associated with frailty, slowed gait, falls, and impaired cognition (19, 20). These associations persist even after accounting for comorbidity burden, suggesting direct medication-related neurocognitive effects. Beyond the known neuropsychiatric effects of certain ART agents like efavirenz, the high burden of non-HIV medications (e.g., benzodiazepines, sedative-hypnotics) — particularly those with anticholinergic properties — is linked to accelerated cognitive decline and increased dementia risk (166–169). These factors underscore the need for a brain health–centered approach to aging PWH, prioritizing deprescribing, minimizing CNS-active agents, and integrating geriatric care (170, 171).
Novel therapeutic approaches toward neuroprotection and HIV cure. Clinical trials of minocycline (172, 173), selegiline (174), and tesamorelin (175), and maraviroc intensification (176), have been unsuccessful so far. The limited success of these prior, primarily antiinflammatory focused trials likely reflects intervention at a late disease stage, absence of molecular stratification, and targeting of downstream inflammation rather than upstream drivers such as persistent viral transcription and glial reprogramming. Modern efforts toward CNS-targeted cure increasingly emphasize combinatorial approaches that reduce reservoir burden (177). Latency-reversing agents remain foundational to “shock-and-kill” strategies (178), but CNS implementation requires caution given distinct reservoir cell types and risks of catastrophic inflammatory neurotoxicity (177, 179). Broadly neutralizing antibodies (bNAbs) provide antiviral and immunomodulatory activity but have limited CNS penetration (180, 181). Gene editing and cell-based approaches remain promising but face challenges in delivery across the blood-brain barrier, off-target effects, and targeting long-lived myeloid reservoirs (181, 182). Emerging, mechanism-based therapies include nanoparticle-based delivery of ART, bNAbs, antioxidants, and antiinflammatory agents (182–185); small-molecule inhibitors of myeloid HIV transcription (186); and CCR2/CCR5 dual inhibitors that target monocyte activation and neuroinvasion (187, 188).
Given that HIV-related brain injury is fundamentally characterized by synaptodendritic injury, the emergence of synaptic regenerative therapies represents a potentially promising therapeutic shift. Pharmaceutical agents that are designed to promote dendritic spine resprouting may help restore synaptic connectivity lost through chronic exposure to viral proteins (189). Similarly, the p75 neurotrophin receptor ligand LM11A-31 has demonstrated preclinical efficacy in HIV models by stabilizing neuronal calcium homeostasis and counteracting pro-apoptotic signaling pathways that contribute to neuropathogenesis (190, 191). By focusing on structural synaptic repair rather than solely on viral suppression, these approaches offer a potential strategy for actively restoring cognitive function, particularly in the aging population of people living with HIV.
-
Looking ahead
While modern ART effectively halts systemic replication, the CNS remains a “smoldering” sanctuary where both intact and defective proviruses drive localized immune activation and microglial dysregulation. As the population of PWH ages, these viral legacy effects intersect with cardiovascular disease, metabolic syndrome, and the cumulative burdens of multimorbidity and polypharmacy. To untangle this multifactorial injury, the field must transition from descriptive neuropsychological thresholds toward a biologically anchored framework that identifies distinct biotypes of injury, paving the road to precision therapeutics. The National Institute of Mental Health Biotypes of CNS Complications formalizes this transition by integrating host and viral genomics, high-field neuroimaging, and deep clinical phenotyping, to identify reproducible biological categories (192). Within this framework, advanced neuroimaging and modern approaches for CSF and neuropathologic analyses are crucial.
Advanced neuroimaging. High-resolution MRI, PET, and emerging imaging tracers have become instrumental to detecting subtle HIV-associated brain abnormalities in ART-treated PWH. Structural and functional MRI identify gray-matter loss, white-matter abnormalities, and disrupted network connectivity, while newer modalities, such as diffusion MRI, quantitative MRI and MR spectroscopy, probe microstructure, inflammation, and glial alterations, particularly in subcortical regions, such as the basal ganglia and corpus callosum (193–196). Functional and perfusion MRI reveal disrupted resting-state connectivity, altered task-related activation, and abnormal cerebral blood flow (197–199). Small-scale, preliminary PET studies using SV2A ligands demonstrate reduced synaptic density correlating with cognitive performance (200), though these findings require validation in larger, well-powered cohorts. Additionally, TSPO tracers localize increased microglial and astrocytic activation (52), while amyloid and tau ligands reveal pathological deposition in the brains of virally suppressed PWH (201). Magnetoencephalography further uncovers aberrant neural oscillatory dynamics (202). Integrated multimodal imaging and machine learning now aim to generate individualized prediction models of NCI (195, 203), representing a transition from descriptive to mechanistic imaging biomarkers.
CSF as a “liquid biopsy.” CSF serves as a vital “window” into the brain. Whereas prior CSF studies primarily focused on the measurement of soluble inflammatory proteins, in an unsuccessful search for a soluble biomarker of HAND, modern studies capitalizing on technical advances in single-cell profiling have begun to reveal the landscape of host immunity in the CNS of PWH through a focus on cells, rather than just soluble proteins, of the CSF. The majority of CSF immune cells, primarily T cells, migrate from the peripheral blood across the blood-CSF barrier at the choroid plexus to provide continuous immune surveillance of the central nervous system. Beyond the systemic circulation, recent research identifies cranial bone marrow as a vital local reservoir that supplies immune cells — particularly myeloid cells and B cells — directly to the meninges and CSF through microscopic vascular channels (204). The meningeal lymphatic system also serves as a specialized conduit that allows for the trafficking and potential recirculation of immune cells between the subarachnoid space and the peripheral lymph nodes (205). However, these alternative sources of CSF cells have not been well studied in the context of HIV infection.
Multimodal platforms integrating transcriptomics, surface proteomics, chromatin accessibility, and histone modification now enable CSF immune cell mapping with a granularity approaching that of brain tissue profiling, allowing for real-time, longitudinal sampling of CSF cells in living people. Coupling these approaches with adaptive immune repertoire sequencing provides high-resolution T cell and B cell receptor clonotypes and insights into clonal architecture, antigen specificity, and trafficking of adaptive immune cells, processes increasingly recognized as central to CNS reservoir persistence (120). scRNA-seq also allows detection of HIV transcripts within CSF-derived cells, supporting models of transcriptionally active but replication-restricted infection driving local immune activation (14, 120), and allowing for the detection of rare, disease-related immune subsets (14, 61).
In addition to single-cell profiling, high-throughput proteomics and metabolomics refine our understanding of neuroinflammation, neuronal injury, and persistent neurometabolic reprogramming across the spectrum of NCI despite viral suppression (11, 44, 206). However, the high dimensionality of these datasets, interindividual variability, and the need for cross-platform harmonization highlight emerging challenges. Future priorities include standardized acquisition and computational harmonization pipelines, larger longitudinal cohorts, and deeper integration with imaging and neuropathology to translate molecular signatures into validated biomarkers and therapeutic targets.
Neuropathology and postmortem brain tissue studies. Long-standing autopsy networks such as the National NeuroHIV Tissue Consortium provide deeply phenotyped tissue (207), while rapid-autopsy programs like Last Gift enable sampling within hours of death, preserving RNA, epigenomic architecture, and viable cells for high-resolution virologic and immunologic analyses (208). Modern single-cell multiomics applied to these tissues have refined our understanding of microglial infection in virally suppressed individuals (48), while multiregion atlas efforts further show substantial regional and interindividual heterogeneity in immune activation and HIV-bearing myeloid states, influenced by comorbid factors such as substance use (209). Spatial approaches alongside tissue-level ART drug quantification further define the regional heterogeneity of the reservoir and the variable CNS penetration of ART (102, 210, 211). Looking forward, key priorities include targeted enrichment methods for single-cell HIV RNA and full-length proviral sequencing, high-resolution and 3D reservoir mapping, and cross-species integration of human and animal model data providing opportunities for preclinical testing and rapid translation of CNS-targeted interventions; fresh CSF-brain paired cohorts to track reservoir dynamics across compartments; and incorporation of unbiased pathogen detection to disentangle HIV-specific injury from comorbid CNS pathology (212).
Achieving the precision-medicine goal requires continued emphasis on high-resolution, human-based studies. While animal models offer valuable insights into HIV neuropathogenesis and generate hypotheses, direct interrogation of the human CNS through longitudinal CSF multiomics and rapid-autopsy programs provides the only definitive means to map the viral reservoir and its epigenomic imprinting on resident glia. These human-centered approaches remain essential for validating the unique innate and adaptive immune programs that are central to chronic neuroinflammation and may not be fully recapitulated in animal models (Table 1).
 Table 1
Table 1Modern human translational approaches to defining HIV persistence and brain injury in the CNS in the ART era
Ultimately, these molecular insights must be viewed through the lens of the global epidemic’s structural realities. Global inequities in neuroHIV diagnosis and management remain substantial. Advanced neuroimaging, CSF biomarker platforms, and neuropathology resources are concentrated in high-income settings, whereas the greatest burden of HIV-associated NCI lies in low- and middle-income countries (213, 214). Early ART initiation and durable suppression clearly mitigate risk of severe HIV-associated NCI, yet disparities in ART access limit these benefits (215). Diagnostic frameworks like the Frascati criteria often misclassify socioeconomic hardships as biological injury (21, 216). Indeed, recent work in South Africa highlights that social determinants of health — including food insecurity, low education, and even structural factors such as lack of indoor bathroom access — exert a major influence on cognitive test performance (21, 216). The finding that social determinants of health can fundamentally dictate cognitive performance serves as a critical warning against the overmedicalization of socioeconomic hardship (213). A rigorous future for neuroHIV research requires a dual commitment: the pursuit of deep, human-derived biological data and the integration of the social context in which these biological processes occur.
-
Conflict of interest
The authors have declared that no conflict of interest exists.
-
Funding support
This work is the result of NIH funding, in whole or in part, and is subject to the NIH Public Access Policy. Through acceptance of this federal funding, the NIH has been given a right to make the work publicly available in PubMed Central.
- NIH R21MH142338, R01DA060493, R01MH134391.
- Swiss National Science Foundation (grant P500PM_221968 to PF).
-
Footnotes
Copyright: © 2026, Filippidis et al. This is an open access article published under the terms of the Creative Commons Attribution 4.0 International License.
Reference information: J Clin Invest. 2026;136(9):e204831. https://doi.org/10.1172/JCI204831.
-
References
- Snider WD, et al. Neurological complications of acquired immune deficiency syndrome: analysis of 50 patients. Ann Neurol. 1983;14(4):403–418.
- Navia BA, et al. The AIDS dementia complex: I. Clinical features. Ann Neurol. 1986;19(6):517–524.
- Heaton RK, et al. HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER Study. Neurology. 2010;75(23):2087–2096.
- Heaton RK, et al. HIV-associated neurocognitive disorders before and during the era of combination antiretroviral therapy: differences in rates, nature, and predictors. J Neurovirol. 2011;17(1):3–16.
- Brew BJ, et al. AIDS dementia complex and HIV-1 brain infection: clinical-virological correlations. Ann Neurol. 1995;38(4):563–570.
- Glass JD, et al. Clinical-neuropathologic correlation in HIV-associated dementia. Neurology. 1993;43(11):2230–2237.
- Simioni S, et al. Cognitive dysfunction in HIV patients despite long-standing suppression of viremia. AIDS. 2010;24(9):1243–1250.
- Crum-Cianflone NF, et al. Low prevalence of neurocognitive impairment in early diagnosed and managed HIV-infected persons. Neurology. 2013;80(4):371–379.
- Lawler K, et al. Neurocognitive impairment among HIV-positive individuals in Botswana: a pilot study. J Int AIDS Soc. 2010;13:15.
- Pumpradit W, et al. Neurocognitive impairment and psychiatric comorbidity in well-controlled human immunodeficiency virus-infected Thais from the 2NN Cohort Study. J Neurovirol. 2010;16(1):76–82.
- Williams ME, et al. Cerebrospinal fluid immune markers and HIV-associated neurocognitive impairments: A systematic review. J Neuroimmunol. 2021;358:577649.
- Eden A, et al. Increased intrathecal immune activation in virally suppressed HIV-1 infected patients with neurocognitive impairment. PLoS One. 2016;11(6):e0157160.
- Jessen Krut J, et al. Biomarker evidence of axonal injury in neuroasymptomatic HIV-1 patients. PLoS One. 2014;9(2):e88591.
- Farhadian SF, et al. HIV viral transcription and immune perturbations in the CNS of people with HIV despite ART. JCI Insight. 2022;7(13):e160267.
- Guaraldi G, et al. Premature age-related comorbidities among HIV-infected persons compared with the general population. Clin Infect Dis. 2011;53(11):1120–1126.
- Kehler DS, et al. Frailty in older people living with HIV: current status and clinical management. BMC Geriatr. 2022;22(1):919.
- Ellis RJ, et al. Higher Comorbidity Burden Predicts Worsening Neurocognitive Trajectories in People with Human Immunodeficiency Virus. Clin Infect Dis. 2022;74(8):1323–1328.
- Lorenz DR, et al. Predictors of transition to frailty in middle-aged and older people with HIV: a prospective cohort study. J Acquir Immune Defic Syndr. 2021;88(5):518–527.
- Greene M, et al. Polypharmacy, drug-drug interactions, and potentially inappropriate medications in older adults with human immunodeficiency virus infection. J Am Geriatr Soc. 2014;62(3):447–453.
- Kosana P, et al. Polypharmacy is associated with slow gait speed and recurrent falls in older people With HIV. Clin Infect Dis. 2024;78(6):1608–1616.
- Dreyer AJ, et al. Psychosocial factors account for a proportion of the difference in cognitive performance between persons with and without HIV. AIDS. 2025;39(4):393–402.
- Chan P, et al. Distribution of human immunodeficiency virus (HIV) ribonucleic acid in cerebrospinal fluid and blood is linked to CD4/CD8 ratio during acute HIV. J Infect Dis. 2018;218(6):937–945.
- Kanmogne GD, et al. HIV-1 gp120 compromises blood-brain barrier integrity and enhances monocyte migration across blood-brain barrier: implication for viral neuropathogenesis. J Cereb Blood Flow Metab. 2007;27(1):123–134.
- Fischer-Smith T, et al. CNS invasion by CD14+/CD16+ peripheral blood-derived monocytes in HIV dementia: perivascular accumulation and reservoir of HIV infection. J Neurovirol. 2001;7(6):528–541.
- Nottet HS, et al. Mechanisms for the transendothelial migration of HIV-1-infected monocytes into brain. J Immunol. 1996;156(3):1284–1295.
- Williams DW, et al. CCR2 on CD14(+)CD16(+) monocytes is a biomarker of HIV-associated neurocognitive disorders. Neurol Neuroimmunol Neuroinflamm. 2014;1(3):e36.
- Chakrabarti L, et al. Early viral replication in the brain of SIV-infected rhesus monkeys. Am J Pathol. 1991;139(6):1273–1280.View this article via: PubMed Google Scholar
- Trillo-Pazos G, et al. Detection of HIV-1 DNA in microglia/macrophages, astrocytes and neurons isolated from brain tissue with HIV-1 encephalitis by laser capture microdissection. Brain Pathol. 2003;13(2):144–154.
- Joseph SB, Swanstrom R. The evolution of HIV-1 entry phenotypes as a guide to changing target cells. J Leukoc Biol. 2018;103(3):421–431.
- Bonner X, et al. Stoichiometry for entry and binding properties of the Env protein of R5 T cell-tropic HIV-1 and its evolutionary variant of macrophage-tropic HIV-1. mBio. 2024;15(4):e0032124.
- Schnell G, et al. HIV-1 replication in the central nervous system occurs in two distinct cell types. PLoS Pathog. 2011;7(10):e1002286.
- Baxter AE, et al. Macrophage infection via selective capture of HIV-1-infected CD4+ T cells. Cell Host Microbe. 2014;16(6):711–721.
- McCrossan M, et al. An immune control model for viral replication in the CNS during presymptomatic HIV infection. Brain. 2006;129(pt 2):503–516.
- Sharma V, et al. Cerebrospinal fluid CD4+ T cell infection in humans and macaques during acute HIV-1 and SHIV infection. PLoS Pathog. 2021;17(12):e1010105.
- Masliah E, et al. Spectrum of human immunodeficiency virus-associated neocortical damage. Ann Neurol. 1992;32(3):321–329.
- Glass JD, et al. Immunocytochemical quantitation of human immunodeficiency virus in the brain: correlations with dementia. Ann Neurol. 1995;38(5):755–762.
- Wiley CA, et al. Neocortical damage during HIV infection. Ann Neurol. 1991;29(6):651–657.
- Schmidbauer M, et al. Multifocal vacuolar leucoencephalopathy: a distinct HIV-associated lesion of the brain. Neuropathol Appl Neurobiol. 1990;16(5):437–443.
- Borjabad A, et al. Significant effects of antiretroviral therapy on global gene expression in brain tissues of patients with HIV-1-associated neurocognitive disorders. PLoS Pathog. 2011;7(9):e1002213.
- Gelman BB, et al. The National NeuroAIDS Tissue Consortium brain gene array: two types of HIV-associated neurocognitive impairment. PLoS One. 2012;7(9):e46178.
- Shapshak P, et al. Independent evolution of HIV type 1 in different brain regions. AIDS Res Hum Retroviruses. 1999;15(9):811–820.
- Chan P, Valcour V. Neurocognition and the Aging Brain in People With HIV: Implications for Screening. Top Antivir Med. 2022;29(5):423–429.View this article via: PubMed Google Scholar
- Letendre SL, et al. Chemokines in cerebrospinal fluid correlate with cerebral metabolite patterns in HIV-infected individuals. J Neurovirol. 2011;17(1):63–69.
- Hu Z, et al. Changes in cerebrospinal fluid proteins across the spectrum of untreated and treated chronic HIV-1 infection. PLoS Pathog. 2024;20(9):e1012470.
- Mrdjen D, et al. High-dimensional single-cell mapping of central nervous system immune cells reveals distinct myeloid subsets in health, aging, and disease. Immunity. 2018;48(2):380–395.
- Utz SG, et al. Early fate defines microglia and non-parenchymal brain macrophage development. Cell. 2020;181(3):557–573.
- Vara-Perez M, Movahedi K. Border-associated macrophages as gatekeepers of brain homeostasis and immunity. Immunity. 2025;58(5):1085–1100.
- Schlachetzki JC, et al. Gene expression and chromatin conformation of microglia in virally suppressed people with HIV. Life Sci Alliance. 2024;7(10):e202402736.
- Garcia-Dominguez M. Relationship of S100 proteins with neuroinflammation. Biomolecules. 2025;15(8):1125.
- Kong W, et al. Neuroinflammation generated by HIV-infected microglia promotes dysfunction and death of neurons in human brain organoids. PNAS Nexus. 2024;3(5):pgae179.
- Coughlin JM, et al. Regional brain distribution of translocator protein using [(11)C]DPA-713 PET in individuals infected with HIV. J Neurovirol. 2014;20(3):219–232.
- Vera JH, et al. Neuroinflammation in treated HIV-positive individuals: A TSPO PET study. Neurology. 2016;86(15):1425–1432.
- Plaza-Jennings AL, et al. HIV integration in the human brain is linked to microglial activation and 3D genome remodeling. Mol Cell. 2022;82(24):4647–4663.
- Chivero ET, et al. HIV-1 tat primes and activates microglial NLRP3 inflammasome-mediated neuroinflammation. J Neurosci. 2017;37(13):3599–3609.
- He X, et al. NLRP3-dependent pyroptosis is required for HIV-1 gp120-induced neuropathology. Cell Mol Immunol. 2020;17(3):283–299.
- Katuri A, et al. Role of the inflammasomes in HIV-associated neuroinflammation and neurocognitive disorders. Exp Mol Pathol. 2019;108:64–72.
- Mamik MK, et al. HIV-1 viral protein R activates NLRP3 inflammasome in microglia: implications for HIV-1 associated neuroinflammation. J Neuroimmune Pharmacol. 2017;12(2):233–248.
- Walsh JG, et al. Rapid inflammasome activation in microglia contributes to brain disease in HIV/AIDS. Retrovirology. 2014;11:35.
- Filippidis P, et al. Shared microglia subtypes in brain and CSF reveal HIV-driven immune dysregulation.Conference on Retroviruses and Opportunistic Infections. February 22-25, 2026; Denver, Colorado, USA.
- Martins-Ferreira R, et al. The Human Microglia Atlas (HuMicA) unravels changes in disease-associated microglia subsets across neurodegenerative conditions. Nat Commun. 2025;16(1):739.
- Farhadian SF, et al. Single-cell RNA sequencing reveals microglia-like cells in cerebrospinal fluid during virologically suppressed HIV. JCI Insight. 2018;3(18):e121718.
- Grauer OM, et al. Neurocognitive decline in HIV patients is associated with ongoing T cell activation in the cerebrospinal fluid. Ann Clin Transl Neurol. 2015;2(9):906–919.
- Amundson B, et al. Distinct cellular immune properties in cerebrospinal fluid are associated with cognition in HIV-infected individuals initiating antiretroviral therapy. J Neuroimmunol. 2020;344:577246.
- Albalawi YA, et al. CD4 dim CD8 bright T cells are inversely associated with neuro-inflammatory markers among people with HIV. AIDS. 2024;38(1):1–7.
- Chan P, et al. Longitudinal CNS and systemic T-lymphocyte and monocyte activation before and after antiretroviral therapy beginning in primary HIV infection. Front Immunol. 2025;16:1531828.
- Schrier RD, et al. Cerebrospinal fluid (CSF) CD8+ T cells that express interferon-gamma contribute to HIV associated neurocognitive disorders (HAND). PLoS One. 2015;10(2):e0116526.
- Kundura L, et al. Low levels of peripheral blood activated and senescent T cells characterize people with HIV-1-associated neurocognitive disorders. Front Immunol. 2023;14:1267564.
- Mitchell BI, et al. Dynamics of peripheral T cell exhaustion and monocyte subpopulations in neurocognitive impairment and brain atrophy in chronic HIV infection. J Neurovirol. 2024;30(5–6):489–499.
- Hart M, et al. Loss of discrete memory B cell subsets is associated with impaired immunization responses in HIV-1 infection and may be a risk factor for invasive pneumococcal disease. J Immunol. 2007;178(12):8212–8220.
- Titanji K, et al. Loss of memory B cells impairs maintenance of long-term serologic memory during HIV-1 infection. Blood. 2006;108(5):1580–1587.
- Trunfio M, et al. Cerebrospinal fluid CXCL13 as candidate biomarker of intrathecal immune activation, IgG synthesis and neurocognitive impairment in people with HIV. J Neuroimmune Pharmacol. 2023;18(1–2):169–182.
- Bonnan M, et al. Compartmentalized intrathecal immunoglobulin synthesis during HIV infection - a model of chronic CNS inflammation? J Neuroimmunol. 2015;285:41–52.
- De Almeida SM, et al. IgG intrathecal synthesis in HIV-associated neurocognitive disorder (HAND) according to the HIV-1 subtypes and pattern of HIV RNA in CNS and plasma compartments. J Neuroimmunol. 2021;355:577542.
- Spatola M, et al. Functional compartmentalization of antibodies in the central nervous system during chronic HIV infection. J Infect Dis. 2022;226(4):738–750.
- Trunfio M, et al. Intrathecal production of anti-Epstein-Barr virus viral capsid antigen IgG is associated with neurocognition and tau proteins in people with HIV. AIDS. 2024;38(4):477–486.
- Cheng D, et al. Elevated cerebrospinal fluid anti-CD4 autoantibody levels in HIV associate with neuroinflammation. Microbiol Spectr. 2022;10(1):e0197521.
- Hawes IA, et al. Viral co-infection, autoimmunity, and CSF HIV antibody profiles in HIV central nervous system escape. J Neuroimmunol. 2023;381:578141.
- Akbay B, et al. Modulation of mTORC1 signaling pathway by HIV-1. Cells. 2020;9(5):1090.
- Cinti A, et al. HIV-1 enhances mTORC1 activity and repositions lysosomes to the periphery by co-opting Rag GTPases. Sci Rep. 2017;7(1):5515.
- Achim CL, et al. Increased accumulation of intraneuronal amyloid beta in HIV-infected patients. J Neuroimmune Pharmacol. 2009;4(2):190–199.
- Ellis RJ, et al. Higher cerebrospinal fluid biomarkers of neuronal injury in HIV-associated neurocognitive impairment. J Neurovirol. 2022;28(3):438–445.
- Green DA, et al. Brain deposition of beta-amyloid is a common pathologic feature in HIV positive patients. AIDS. 2005;19(4):407–411.
- Swinton MK, et al. Mitochondrial biogenesis is altered in HIV+ brains exposed to ART: Implications for therapeutic targeting of astroglia. Neurobiol Dis. 2019;130:104502.
- Teodorof-Diedrich C, Spector SA. Human immunodeficiency virus type 1 gp120 and Tat induce mitochondrial fragmentation and incomplete mitophagy in human neurons. J Virol. 2018;92(22):e00993-18.
- Thangaraj A, et al. HIV-1 TAT-mediated microglial activation: role of mitochondrial dysfunction and defective mitophagy. Autophagy. 2018;14(9):1596–1619.
- Abdel-Haq R, et al. Microbiome-microglia connections via the gut-brain axis. J Exp Med. 2019;216(1):41–59.
- Byrnes SJ, et al. Chronic immune activation and gut barrier dysfunction is associated with neuroinflammation in ART-suppressed SIV+ rhesus macaques. PLoS Pathog. 2023;19(3):e1011290.
- Ma Q, et al. Impact of microbiota on central nervous system and neurological diseases: the gut-brain axis. J Neuroinflammation. 2019;16(1):53.
- Hong S, Banks WA. Role of the immune system in HIV-associated neuroinflammation and neurocognitive implications. Brain Behav Immun. 2015;45:1–12.
- Jadhav S, Nema V. HIV-associated neurotoxicity: the interplay of host and viral proteins. Mediators Inflamm. 2021;2021:1267041.
- Tavasoli A, et al. Increasing neuroinflammation relates to increasing neurodegeneration in people with HIV. Viruses. 2023;15(9):1835.
- van Paassen PM, et al. HIV-specific CD8+ T-cell proliferative response 24 weeks after early antiretroviral therapy initiation is associated with the subsequent reduction in the viral reservoir. eLife Sciences Publications, Ltd; 2025.
- Bruner KM, et al. A quantitative approach for measuring the reservoir of latent HIV-1 proviruses. Nature. 2019;566(7742):120–125.
- Moar P, et al. The latent HIV reservoir: current advances in genetic sequencing approaches. mBio. 2023;14(5):e0134423.
- Sadri G, et al. Development and validation of HIV SMRTcap for the characterization of HIV-1 reservoirs across tissues and subtypes. PLoS Pathog. 2026;22(1):e1013171.
- Cochrane CR, et al. Intact HIV proviruses persist in the brain despite viral suppression with ART. Ann Neurol. 2022;92(4):532–544.
- Donoso M, et al. Identification, quantification, and characterization of HIV-1 reservoirs in the human brain. Cells. 2022;11(15):2379.
- Sun W, et al. Persistence of intact HIV-1 proviruses in the brain during antiretroviral therapy. Elife. 2023;12:RP89837.
- Zhang Z, et al. The identification of intact HIV proviral DNA from human cerebrospinal fluid. Neurotherapeutics. 2024;21(4):e00373.
- Gabuzda D, et al. Intact proviral DNA analysis of the brain viral reservoir and relationship to neuroinflammation in people with HIV on suppressive antiretroviral therapy. Viruses. 2023;15(4):1009.
- Agsalda-Garcia M, et al. Different levels of HIV DNA copy numbers in cerebrospinal fluid cellular subsets. J Health Care Poor Underserved. 2013;24(4 suppl):8–16.
- Ko A, et al. Macrophages but not astrocytes harbor HIV DNA in the Brains of HIV-1-infected aviremic individuals on suppressive antiretroviral therapy. J Neuroimmune Pharmacol. 2019;14(1):110–119.
- Spudich S, et al. Persistent HIV-infected cells in cerebrospinal fluid are associated with poorer neurocognitive performance. J Clin Invest. 2019;129(8):3339–3346.
- Byrnes SJ, et al. Neuroinflammation associated with proviral DNA persists in the brain of virally suppressed people with HIV. Front Immunol. 2025;16:1570692.
- Nuhn MM, et al. Microglia exhibit a unique intact HIV reservoir in human postmortem brain tissue. Viruses. 2025;17(4):467.
- Tang Y, et al. Brain microglia serve as a persistent HIV reservoir despite durable antiretroviral therapy. J Clin Invest. 2023;133(12):e167417.
- Liu Z, et al. Subtype C HIV-1 reservoirs throughout the body in ART-suppressed individuals. JCI Insight. 2022;7(20):e162604.
- Albalawi YA, et al. CD4dim CD8bright T cells home to the brain and mediate HIV neuroinvasion. J Virol. 2022;96(15):e0080422.
- Churchill MJ, et al. Use of laser capture microdissection to detect integrated HIV-1 DNA in macrophages and astrocytes from autopsy brain tissues. J Neurovirol. 2006;12(2):146–152.
- Lutgen V, et al. HIV infects astrocytes in vivo and egresses from the brain to the periphery. PLoS Pathog. 2020;16(6):e1008381.
- Malik S, et al. HIV infection of astrocytes compromises inter-organelle interactions and inositol phosphate metabolism: A potential mechanism of bystander damage and viral reservoir survival. Prog Neurobiol. 2021;206:102157.
- Valdebenito S, et al. Astrocytes are HIV reservoirs in the brain: A cell type with poor HIV infectivity and replication but efficient cell-to-cell viral transfer. J Neurochem. 2021;158(2):429–443.
- Li GH, et al. Cell-to-cell contact facilitates HIV transmission from lymphocytes to astrocytes via CXCR4. AIDS. 2015;29(7):755–766.
- Dufour C, et al. Near full-length HIV sequencing in multiple tissues collected postmortem reveals shared clonal expansions across distinct reservoirs during ART. Cell Rep. 2023;42(9):113053.
- Imamichi H, et al. Defective HIV-1 proviruses produce viral proteins. Proc Natl Acad Sci U S A. 2020;117(7):3704–3710.
- Oliveira MF, et al. Evaluation of archival HIV DNA in brain and lymphoid tissues. J Virol. 2023;97(6):e0054323.
- Kuniholm J, et al. Intragenic proviral elements support transcription of defective HIV-1 proviruses. PLoS Pathog. 2021;17(12):e1009982.
- Jamal Eddine J, et al. HIV transcription persists in the brain of virally suppressed people with HIV. PLoS Pathog. 2024;20(8):e1012446.
- Suzuki K, et al. Elevation of cell-associated HIV-1 transcripts in CSF CD4+ T cells, despite effective antiretroviral therapy, is linked to brain injury. Proc Natl Acad Sci U S A. 2022;119(48):e2210584119.
- Wang M, et al. HIV-1-infected T cell clones are shared across cerebrospinal fluid and blood during ART. JCI Insight. 2024;9(7):e176208.
- Elizaldi SR, et al. Deep analysis of CD4 T cells in the rhesus CNS during SIV infection. PLoS Pathog. 2023;19(12):e1011844.
- DeMarino C, et al. HIV-1 RNA in extracellular vesicles is associated with neurocognitive outcomes. Nat Commun. 2024;15(1):4391.
- Agbey C, et al. Expression of HIV envelope protein in the human central nervous system. AIDS. 2025;39(7):788–797.
- Henderson LJ, et al. Presence of Tat and transactivation response element in spinal fluid despite antiretroviral therapy. AIDS. 2019;33(suppl 2):S145–S157.
- Shrestha J, et al. HIV-1 gp120 impairs spatial memory through cyclic AMP response element-binding protein. Front Aging Neurosci. 2022;14:811481.
- Toggas SM, et al. Central nervous system damage produced by expression of the HIV-1 coat protein gp120 in transgenic mice. Nature. 1994;367(6459):188–193.
- Zhang X, et al. HIV gp120-induced neuroinflammation potentiates NMDA receptors to overcome basal suppression of inhibitory synapses by p38 MAPK. J Neurochem. 2019;148(4):499–515.
- Fitting S, et al. Synaptic dysfunction in the hippocampus accompanies learning and memory deficits in human immunodeficiency virus type-1 Tat transgenic mice. Biol Psychiatry. 2013;73(5):443–453.
- Scuderi G, et al. Multifaceted role of Nef in HIV-associated neurocognitive disorder: histopathological alterations and underlying mechanisms. Brain Sci. 2025;15(9):987.
- Raymond AD, et al. HIV Type 1 Nef is released from infected cells in CD45(+) microvesicles and is present in the plasma of HIV-infected individuals. AIDS Res Hum Retroviruses. 2011;27(2):167–178.
- Hu G, et al. Exosome-mediated shuttling of microRNA-29 regulates HIV Tat and morphine-mediated neuronal dysfunction. Cell Death Dis. 2012;3(8):e381.
- Lenassi M, et al. HIV Nef is secreted in exosomes and triggers apoptosis in bystander CD4+ T cells. Traffic. 2010;11(1):110–122.
- Kim HJ, et al. Human immunodeficiency virus protein Tat induces synapse loss via a reversible process that is distinct from cell death. J Neurosci. 2008;28(48):12604–12613.
- Li W, et al. NMDA receptor activation by HIV-Tat protein is clade dependent. J Neurosci. 2008;28(47):12190–12198.
- Hu XT. HIV-1 Tat-mediated calcium dysregulation and neuronal dysfunction in vulnerable brain regions. Curr Drug Targets. 2016;17(1):4–14.
- Wheeler D, et al. Tumor necrosis factor-alpha-induced neutral sphingomyelinase-2 modulates synaptic plasticity by controlling the membrane insertion of NMDA receptors. J Neurochem. 2009;109(5):1237–1249.
- Green MV, Thayer SA. NMDARs adapt to neurotoxic HIV protein Tat downstream of a GluN2A-ubiquitin ligase signaling pathway. J Neurosci. 2016;36(50):12640–12649.
- Zhou BY, et al. Astrocyte activation and dysfunction and neuron death by HIV-1 Tat expression in astrocytes. Mol Cell Neurosci. 2004;27(3):296–305.
- Wang Z, et al. Reduced expression of glutamate transporter EAAT2 and impaired glutamate transport in human primary astrocytes exposed to HIV-1 or gp120. Virology. 2003;312(1):60–73.
- Cisneros IE, Ghorpade A. HIV-1, methamphetamine and astrocyte glutamate regulation: combined excitotoxic implications for neuro-AIDS. Curr HIV Res. 2012;10(5):392–406.
- McMullan HM, et al. Antiretroviral drugs from multiple classes induce loss of excitatory synapses between hippocampal neurons in culture. Front Pharmacol. 2024;15:1369757.
- Bahri MA, et al. Measuring brain synaptic vesicle protein 2A with positron emission tomography and [18F]UCB-H. Alzheimers Dement (N Y). 2017;3(4):481–486.
- Nightingale S, et al. Cognitive impairment in people living with HIV: consensus recommendations for a new approach. Nat Rev Neurol. 2023;19(7):424–433.
- Aung HL, et al. Is there any evidence of premature, accentuated and accelerated aging effects on neurocognition in people living with HIV? A systematic review. AIDS Behav. 2021;25(3):917–960.
- Aung HL, et al. Abnormal cognitive aging in people with HIV: evidence from data integration between two countries’ cohort studies. AIDS. 2022;36(8):1171–1179.
- Heaton RK, et al. Neurocognitive change in the era of HIV combination antiretroviral therapy: the longitudinal CHARTER study. Clin Infect Dis. 2015;60(3):473–480.
- Lam JO, et al. Comparison of dementia risk after age 50 between individuals with and without HIV infection. AIDS. 2021;35(5):821–828.
- Lam JO, et al. Comparison of dementia incidence and prevalence between individuals with and without HIV infection in primary care from 2000 to 2016. AIDS. 2022;36(3):437–445.
- Saloner R, et al. Effects of comorbidity burden and age on brain integrity in HIV. AIDS. 2019;33(7):1175–1185.
- Benjamin LA, et al. HIV, antiretroviral treatment, hypertension, and stroke in Malawian adults: A case-control study. Neurology. 2016;86(4):324–333.
- Moulignier A, et al. Silent cerebral small-vessel disease is twice as prevalent in middle-aged individuals with well-controlled, combination antiretroviral therapy-treated human immunodeficiency virus (HIV) than in HIV-uninfected individuals. Clin Infect Dis. 2018;66(11):1762–1769.
- Gutierrez J, et al. Cerebrovascular contributions to neurocognitive disorders in people living with HIV. J Acquir Immune Defic Syndr. 2021;88(1):79–85.
- McIntosh EC, et al. HIV, Vascular risk factors, and cognition in the combination antiretroviral therapy era: a systematic review and meta-analysis. J Int Neuropsychol Soc. 2021;27(4):365–381.
- Becker JT, et al. Vascular risk factors, HIV serostatus, and cognitive dysfunction in gay and bisexual men. Neurology. 2009;73(16):1292–1299.
- Saloner R, et al. Plasma biomarkers of vascular dysfunction uniquely relate to a vascular-risk profile of neurocognitive deficits in virally-suppressed adults with HIV. Brain Behav Immun Health. 2022;26:100560.
- Carrico AW, et al. Substance-associated elevations in monocyte activation among methamphetamine users with treated HIV infection. AIDS. 2018;32(6):767–771.
- Chilunda V, et al. The impact of substance abuse on HIV-mediated neuropathogenesis in the current ART era. Brain Res. 2019;1724:146426.
- Haorah J, et al. Neurological impact of HIV/AIDS and substance use alters brain function and structure. Front Med (Lausanne). 2024;11:1505440.
- Gianella S, Letendre S. Cytomegalovirus and HIV: a dangerous Pas de Deux. J Infect Dis. 2016;214 Suppl 2(suppl 2):S67–S74.
- Letendre S, et al. Higher anti-cytomegalovirus immunoglobulin G concentrations are associated with worse neurocognitive performance during suppressive antiretroviral therapy. Clin Infect Dis. 2018;67(5):770–777.
- Cassol E, et al. Applications and limitations of inflammatory biomarkers for studies on neurocognitive impairment in HIV infection. J Neuroimmune Pharmacol. 2013;8(5):1087–1097.
- Lupia T, et al. Presence of Epstein-Barr virus DNA in cerebrospinal fluid is associated with greater HIV RNA and inflammation. AIDS. 2020;34(3):373–380.
- Licht-Murava A, et al. Astrocytic TDP-43 dysregulation impairs memory by modulating antiviral pathways and interferon-inducible chemokines. Sci Adv. 2023;9(16):eade1282.
- Pungitore S, et al. Computable phenotypes for post-acute sequelae of SARS-CoV-2: a national COVID cohort collaborative analysis. AMIA Annu Symp Proc. 2023;2023:589–598.View this article via: PubMed Google Scholar
- Xie L, et al. Sleep drives metabolite clearance from the adult brain. Science. 2013;342(6156):373–377.
- Fumaz CR, et al. Long-term neuropsychiatric disorders on efavirenz-based approaches: quality of life, psychologic issues, and adherence. J Acquir Immune Defic Syndr. 2005;38(5):560–565.
- Campbell N, et al. The cognitive impact of anticholinergics: a clinical review. Clin Interv Aging. 2009;4:225–233.View this article via: PubMed Google Scholar
- Fox C, et al. Anticholinergic medication use and cognitive impairment in the older population: the medical research council cognitive function and ageing study. J Am Geriatr Soc. 2011;59(8):1477–1483.
- Jakeman B, et al. Anticholinergic medication use in elderly people living with HIV and self-reported neurocognitive impairment: a prospective cohort study. J Antimicrob Chemother. 2022;77(2):492–499.
- Edelman EJ, et al. Polypharmacy in HIV: recent insights and future directions. Curr Opin HIV AIDS. 2020;15(2):126–133.
- Justice AC, Tate JP. Ageing with and without HIV: will advanced age bring equity or greater disparity? J Int AIDS Soc. 2019;22(9):e25400.
- Nakasujja N, et al. Randomized trial of minocycline in the treatment of HIV-associated cognitive impairment. Neurology. 2013;80(2):196–202.
- Sacktor N, et al. Minocycline treatment for HIV-associated cognitive impairment: results from a randomized trial. Neurology. 2011;77(12):1135–1142.
- Schifitto G, et al. Selegiline and oxidative stress in HIV-associated cognitive impairment. Neurology. 2009;73(23):1975–1981.
- Ellis RJ, et al. Effects of tesamorelin on neurocognitive impairment in persons with HIV and abdominal obesity. J Infect Dis. 2025;231(5):1230–1238.
- Letendre SL, et al. Antiretroviral therapy intensification for neurocognitive impairment in human immunodeficiency virus. Clin Infect Dis. 2023;77(6):866–874.
- Saeb S, et al. Targeting and eradicating latent CNS reservoirs of HIV-1: Original strategies and new models. Biochem Pharmacol. 2023;214:115679.
- Nuhn MM, et al. Shock and kill within the CNS: A promising HIV eradication approach? J Leukoc Biol. 2022;112(5):1297–1315.
- Marban C, et al. Targeting the brain reservoirs: toward an HIV Cure. Front Immunol. 2016;7:397.
- Lorin V, et al. Antibody neutralization of HIV-1 crossing the blood-brain barrier. mBio. 2020;11(5):e02424-20.
- Mastrangelo A, et al. Strategies to target the central nervous system HIV reservoir. Curr Opin HIV AIDS. 2024;19(3):133–140.
- Said N, Venketaraman V. Neuroinflammation, blood-brain barrier, and HIV reservoirs in the CNS: an in-depth exploration of latency mechanisms and emerging therapeutic strategies. Viruses. 2025;17(4):572.
- Gagliardi S, et al. Oxidative stress in HIV-associated neurodegeneration: mechanisms of pathogenesis and therapeutic targets. Int J Mol Sci. 2025;26(14):6724.
- Wen J, et al. Improved delivery of broadly neutralizing antibodies by nanocapsules suppresses SHIV infection in the CNS of infant rhesus macaques. PLoS Pathog. 2021;17(7):e1009738.
- Surnar B, et al. Brain-accumulating nanoparticles for assisting astrocytes to reduce human immunodeficiency virus and drug abuse-induced neuroinflammation and oxidative stress. ACS Nano. 2021;15(10):15741–15753.
- Alamer E, et al. Epigenetic suppression of HIV in myeloid cells by the BRD4-Selective Small Molecule Modulator ZL0580. J Virol. 2020;94(11):e01880-19.
- D’Antoni ML, et al. Improved cognitive performance and reduced monocyte activation in virally suppressed chronic hiv after dual CCR2 and CCR5 antagonism. J Acquir Immune Defic Syndr. 2018;79(1):108–116.
- Veenstra M, et al. Mechanisms of CNS viral seeding by HIV+ CD14+ CD16+ monocytes: establishment and reseeding of viral reservoirs contributing to HIV-associated neurocognitive disorders. mBio. 2017;8(5):e01280-17.
- Trujillo-Estrada L, et al. SPG302 reverses synaptic and cognitive deficits without altering amyloid or Tau pathology in a transgenic model of Alzheimer’s Disease. Neurotherapeutics. 2021;18(4):2468–2483.
- Meeker RB, et al. Novel p75 neurotrophin receptor ligand stabilizes neuronal calcium, preserves mitochondrial movement and protects against HIV associated neuropathogenesis. Exp Neurol. 2016;275 Pt 1(0 1):182–198.
- Meeker RB, et al. Suppression of immunodeficiency virus-associated neural damage by the p75 neurotrophin receptor ligand, LM11A-31, in an in vitro feline model. J Neuroimmune Pharmacol. 2012;7(2):388–400.
- Joseph SB, et al. Biotypes of central nervous system complications in people with human immunodeficiency virus: virology, immunology, and neuropathology. J Infect Dis. 2023;227(suppl 1):S3–S15.
- Chaganti J, Brew BJ. MR spectroscopy in HIV associated neurocognitive disorder in the era of cART: a review. AIDS Res Ther. 2021;18(1):65.
- O’Connor EE, et al. Reliability of white matter microstructural changes in HIV infection: meta-analysis and confirmation. AJNR Am J Neuroradiol. 2017;38(8):1510–1519.
- O’Connor EE, et al. Imaging of brain structural and functional effects in people with human immunodeficiency virus. J Infect Dis. 2023;227(suppl 1):S16–S29.
- Strain JF, et al. Diffusion basis spectral imaging detects ongoing brain inflammation in virologically well-controlled HIV+ patients. J Acquir Immune Defic Syndr. 2017;76(4):423–430.
- Egbert AR, et al. Age and HIV effects on resting state of the brain in relationship to neurocognitive functioning. Behav Brain Res. 2018;344:20–27.
- Ernst T, et al. Abnormal brain activation on functional MRI in cognitively asymptomatic HIV patients. Neurology. 2002;59(9):1343–1349.
- Petersen KJ, et al. Accelerated brain aging and cerebral blood flow reduction in persons with human immunodeficiency virus. Clin Infect Dis. 2021;73(10):1813–1821.
- Weiss JJ, et al. Preliminary in vivo evidence of reduced synaptic density in human immunodeficiency virus (HIV) despite antiretroviral therapy. Clin Infect Dis. 2021;73(8):1404–1411.
- Sinharay S, Hammoud DA. Brain PET imaging: value for understanding the pathophysiology of HIV-associated neurocognitive disorder (HAND). Curr HIV/AIDS Rep. 2019;16(1):66–75.
- Wilson TW, et al. Aberrant brain dynamics in neuroHIV: Evidence from magnetoencephalographic (MEG) imaging. Prog Mol Biol Transl Sci. 2019;165:285–320.View this article via: PubMed Google Scholar
- Li X, et al. The individualized prediction of neurocognitive function in people living with HIV based on clinical and multimodal connectome data. IEEE J Biomed Health Inform. 2023;27(4):2094–2104.View this article via: PubMed Google Scholar
- Mazzitelli JA, et al. Skull bone marrow channels as immune gateways to the central nervous system. Nat Neurosci. 2023;26(12):2052–2062.
- Louveau A, et al. CNS lymphatic drainage and neuroinflammation are regulated by meningeal lymphatic vasculature. Nat Neurosci. 2018;21(10):1380–1391.
- Williams ME, et al. Proteomics and metabolomics of HIV-associated neurocognitive disorders: A systematic review. J Neurochem. 2021;157(3):429–449.
- Morgello S, et al. The National NeuroAIDS Tissue Consortium: a new paradigm in brain banking with an emphasis on infectious disease. Neuropathol Appl Neurobiol. 2001;27(4):326–335.
- Riggs PK, et al. Lessons for understanding central nervous system HIV reservoirs from the last gift program. Curr HIV/AIDS Rep. 2022;19(6):566–579.
- Ament SA, et al. The single-cell opioid responses in the context of HIV (SCORCH) consortium. Mol Psychiatry. 2024;29(12):3950–3961.
- Carstens JL, et al. Spatial multiplexing and omics. Nat Rev Methods Primers. 2024;4(1):54.
- Ferrara M, et al. Antiretroviral drug concentrations in brain tissue of adult decedents. AIDS. 2020;34(13):1907–1914.
- Filippidis P, Corley MJ. Single cell analyses of the HIV reservoir in the CNS and CSF: recent insights and implications. Curr Opin HIV AIDS. 2025;20(5):493–501.
- Olajide T, et al. HIV-associated neurocognitive disorders in Africa: challenges, peculiarities, and future directions. AIDS Res Ther. 2024;21(1):88.
- Zenebe Y, et al. Worldwide occurrence of HIV-associated neurocognitive disorders and its associated factors: a systematic review and meta-analysis. Front Psychiatry. 2022;13:814362.
- Nweke M, et al. Meta-analysis of factors affecting prevalence estimates of HIV-associated neurocognitive disorder in sub-Saharan Africa. S Afr J Sci. 2021;117(9/10):1–10.View this article via: CrossRef Google Scholar
- Dreyer AJ, et al. Cognitive performance in a South African cohort of people with HIV and comorbid major depressive disorder. J Neurovirol. 2022;28(4–6):537–551.
-
Version history
- Version 1 (May 1, 2026): Electronic publication
Article tools
-
View PDF

- Download citation information
- Send a comment
- Terms of use
- Standard abbreviations
- Need help? Email the journal
Metrics
Go to
- Top
- Abstract
- Introduction
- HIV’s impact on the CNS before treatment
- HIV in the brain after ART: host immunity
- HIV in the brain after ART: viral products
- Clinical challenges and frameworks: HIV, aging, and comorbidities
- Looking ahead
- Conflict of interest
- Funding support
- Footnotes
- References
- Version history



Copyright © 2026 American Society for Clinical Investigation
ISSN: 0021-9738 (print), 1558-8238 (online)



