Abstract
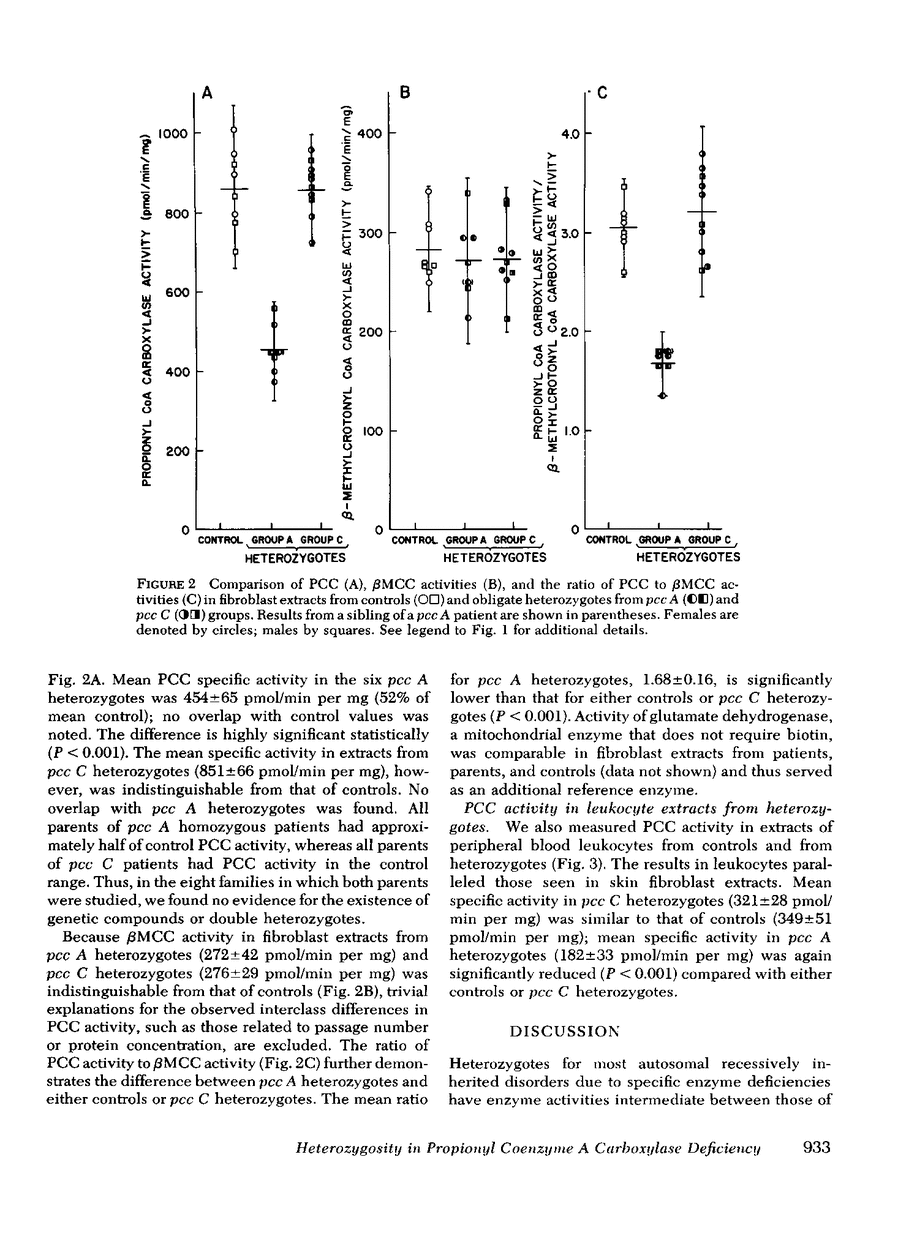
We measured propionyl coenzyme A carboxylase (PCC) activity in extracts of skin fibroblasts and peripheral blood leukocytes from controls and obligate heterozygotes for PCC deficiency. 6 heterozygotes were from the pcc A complementation group; 12 were from the other major complementation group, designated pcc C. Mean PCC activity in fibroblast extracts from pcc A heterozygotes was 52% of that in controls, whereas mean PCC activity in pcc C heterozygotes was indistinguishable from that of controls. Similar results were obtained with extracts of peripheral blood leukocytes. In none of eight families (three pcc A and five pcc C) in which PCC activity was studied in both parents of an affected child were significant intrafamilial differences observed. The activities of two other mitochondrial enzymes (β-methyl-crotonyl CoA carboxylase and glutamate dehydrogenase) were comparable in controls and both groups of heterozygotes. Whereas the data from pcc A heterozygotes are consistent with expected gene dosage effects, those from pcc C heterozygotes are not. Inasmuch as mammalian PCC is a large molecular weight tetramer, each protomer of which is probably composed of two nonidentical subunits, the latter results are most consistent with unbalanced rates of synthesis and(or) degradation of the two subunits in normal cells with compensatory balancing in pcc C heterozygotes.
Authors
Barry Wolf, Leon E. Rosenberg
Other pages:
| 931 | 932 | 933 | 934 | 935 | 936 |